Debulking of topoisomerase DNA-protein crosslinks (TOP-DPC) by the proteasome, non-proteasomal and non-proteolytic pathways
- PMID: 32674013
- PMCID: PMC9210512
- DOI: 10.1016/j.dnarep.2020.102926
Debulking of topoisomerase DNA-protein crosslinks (TOP-DPC) by the proteasome, non-proteasomal and non-proteolytic pathways
Abstract
Topoisomerases play a pivotal role in ensuring DNA metabolisms during replication, transcription and chromosomal segregation. To manage DNA topology, topoisomerases generate break(s) in the DNA backbone by forming transient enzyme-DNA cleavage complexes (TOPcc) with phosphotyrosyl linkages between DNA ends and topoisomerase catalytic tyrosyl residues. Topoisomerases have been identified as the cellular targets of a variety of anti-cancer drugs (e.g. topotecan, irinotecan, etoposide and doxorubicin, and antibiotics (e.g. ciprofloxacin and levofloxacin). These drugs, as well as other exogenous and endogenous agents, convert the transient TOPcc into persistent TOPcc, which we refer to as topoisomerase DNA-protein crosslinks (TOP-DPC) that challenge genome integrity and lead to cell death if left unrepaired. Proteolysis of the bulky protein component of TOP-DPC (debulking) is a poorly understood repair process employed across eukaryotes. TOP-DPC proteolysis can be achieved either by the ubiquitin-proteasome pathway (UPP) or by non-proteasomal proteases, which are typified by the metalloprotease SPRTN/WSS1. Debulking of TOP-DPC exposes the phosphotyrosyl bonds, hence enables tyrosyl-DNA phosphodiesterases (TDP1 and TDP2) to access and cleave the bonds. In this review, we focus on current knowledge of the protease pathways for debulking TOP-DPC and highlighting recent advances in understanding the mechanisms regulating the proteolytic repair pathways. We also discuss the avenues that are being exploited to target the proteolytic repair pathways for improving the clinical outcome of topoisomerase inhibitors.
Keywords: DNA-protein crosslinks; SPRTN; SUMOylation; Topoisomerases; Ubiquitin-proteasome pathway.
Published by Elsevier B.V.
Conflict of interest statement
Declaration of Competing Interest
The authors declare that there is no conflict of interest.
Figures


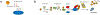

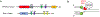
References
-
- Stingele J, Bellelli R, Boulton SJ, Mechanisms of DNA-protein crosslink repair, Nat. Rev. Mol. Cell Biol 18 (2017) 563–573. - PubMed
-
- Ide H, Nakano T, Salem AMH, Shoulkamy MI, DNA-protein cross-links: formidable challenges to maintaining genome integrity, DNA Repair (Amst) 71 (2018) 190–197. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

