Field Study on the Immunological Response and Protective Effect of a Licensed Autogenous Vaccine to Control Streptococcus suis Infections in Post-Weaned Piglets
- PMID: 32674276
- PMCID: PMC7565864
- DOI: 10.3390/vaccines8030384
Field Study on the Immunological Response and Protective Effect of a Licensed Autogenous Vaccine to Control Streptococcus suis Infections in Post-Weaned Piglets
Abstract
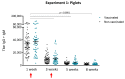
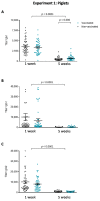
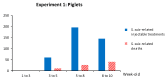
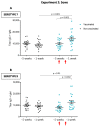
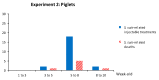
Streptococcus suis is one of the most important bacterial pathogens in weaned piglets and responsible for serious economic losses to the swine industry. Currently, mostly autogenous vaccines composed of killed bacteria (bacterins) are available. However, immunological and protective data from field studies are missing. We report for the first time a comparative field study on the immunological response induced by an autogenous vaccine applied to either piglets or sows in a farm with recurrent S. suis problems. (I) Piglets from non-vaccinated sows received an autogenous bacterin during the first week and at three weeks of age. (II) Sows received the vaccine at five and three weeks pre-farrowing and piglets were non-vaccinated. Levels, isotype profile and opsonophagocytosis capacity of the serum antibodies induced by vaccination were evaluated. Vaccination of piglets failed to induce an active immune response. Vaccination of sows induced a significant increase in anti-S. suis antibodies, mainly composed of IgG1. However, isotype switching was modulated by the S. suis serotype included in the vaccine formulation. Despite this antibody increase in vaccinated sows, transfer of maternal immunity to piglets was not different from the control group (i.e., piglets from non-vaccinated sows). Notably, levels of maternal antibodies in piglets were already very high with marked opsonophagocytosis capacity at one week of age, independently of the vaccination program. However, their levels decreased by three weeks of age, indicating possible absence of antibodies in the post-weaning high-risk period. These observations correlated with lack of clinical protection in the farm. Overall, a piglet or a sow vaccination program herein mostly failed to induce lasting protection in nursery piglets. An improvement of vaccine formulation or an optimized program may be required.
Keywords: Streptococcus suis; autogenous bacterin; field; infection; pigs; vaccine.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures






References
-
- Gottschalk M., Segura M. Streptococcosis. In: Zimmerman J.J., Karriker L.A., Ramirez A., Schwartz K.J., Stevenson G.W., Zhang J., editors. Diseases of Swine. 11th ed. Wiley-Blackwell; West Sussex, UK: 2019. pp. 934–951.
-
- Goyette-Desjardins G., Auger J.P., Xu J., Segura M., Gottschalk M. Streptococcus suis, an important pig pathogen and emerging zoonotic agent—An update on the worldwide distribution based on serotyping and sequence typing. Emerg. Microbes. Infect. 2014;3:e45. doi: 10.1038/emi.2014.45. - DOI - PMC - PubMed
Grants and funding
- IA119059/Programme Innov'Action agroalimentaire of the Ministère de l'Agriculture, des Pêcheries et de l'Alimentation (MAPAQ)
- 342150/Natural Sciences and Engineering Research Council of Canada (NSERC)
- 04435/Natural Sciences and Engineering Research Council of Canada (NSERC)
- RS-170946/Fonds de recherche du Québec-Nature et technologies (FRQNT)
LinkOut - more resources
Full Text Sources

