Differences in Real-World Health and Economic Outcomes Among Patients with COPD Treated with Combination Tiotropium/Olodaterol Versus Triple Therapy
- PMID: 32678719
- PMCID: PMC10390943
- DOI: 10.18553/jmcp.2020.20159
Differences in Real-World Health and Economic Outcomes Among Patients with COPD Treated with Combination Tiotropium/Olodaterol Versus Triple Therapy
Abstract
Background: The 2018 Global Initiative for Chronic Obstructive Lung Disease (GOLD) recommends combination long-acting muscarinic antagonists/long-acting beta2-agonists (LAMA + LABA) as preferred maintenance therapy for patients with symptomatic chronic obstructive lung disease (COPD) after monotherapy and stepping up to triple therapy (TT; LAMA + LABA + inhaled corticosteroids [ICS]) in case of further exacerbations. Restrictions on TT recommendations have primarily been driven by higher pneumonia risk associated with regular ICS use. Evidence suggests that TT is overprescribed, which may affect economic and clinical outcomes.
Objective: To compare health plan-paid costs, COPD exacerbations, and pneumonia diagnoses among patients newly treated with a LAMA + LABA regimen composed of tiotropium (TIO) + olodaterol (OLO) in a fixed-dose combination inhaler (TIO + OLO) or TT in a U.S. Medicare Advantage Part D insured population.
Methods: This retrospective study identified COPD patients aged ≥ 40 years who were initiating TIO + OLO or TT (index regimen) between January 1, 2014, and March 31, 2018, from a national administrative claims database. Continuous insurance coverage for 12 months pretreatment (baseline) and ≥ 30 days posttreatment (follow-up) was required. Patients were followed until the earliest of study end (May 31, 2018), discontinuation of index regimen (≥ 60-day gap in index regimen coverage), switch to a different regimen, or health plan disenrollment. Before analysis of outcomes, TIO + OLO and TT patients were 1:1 propensity score-matched on baseline demographics, comorbidities, COPD medication use, medical resource use, and costs. Cohort differences in post-match outcomes were assessed by Wald Z-test (annualized costs) and Kaplan-Meier method (time to first COPD exacerbation and pneumonia diagnosis).
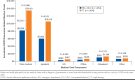
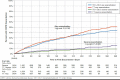
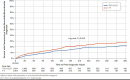
Results: After matching, each cohort had 1,454 patients who were well balanced on baseline characteristics. Compared with TT, the TIO + OLO cohort incurred $7,041 (41.1%) lower mean COPD-related total costs ($10,094 vs. $17,135; P < 0.001); cohort differences in the medical component ($3,666 lower for TIO + OLO) were driven by lower mean acute inpatient costs ($3,053 lower for TIO + OLO). Combined mean COPD plus pneumonia-related medical costs were $5,212 (39.0%) lower for TIO + OLO versus TT ($8,209 vs. $13,421; P = 0.006), and total mean all-cause costs were $9,221 (30.4%) lower for TIO + OLO versus TT ($21,062 vs. $30,283; P < 0.001). Kaplan-Meier analysis found longer time to first severe COPD exacerbation (P = 0.020) and first pneumonia diagnosis (P = 0.002) for TIO + OLO versus TT and a lower percentage of TIO + OLO patients experiencing these events (severe COPD exacerbation: 9.0% vs. 16.1%; pneumonia: 14.5% vs. 19.3%). A secondary analysis, which expanded the TIO + OLO cohort to include any LAMA + LABA regimen, had similar findings for all outcomes.
Conclusions: COPD patients initiating TIO + OLO incurred lower costs to health plans and experienced fewer COPD exacerbation and pneumonia events relative to TT. These findings provide important real-world economic and clinical insight into the GOLD recommendations for TIO + OLO and LAMA + LABA therapy. The study findings also indicate the continued inconsistency between the recommendations and real-world clinical practices pertaining to TT.
Disclosures: This study was sponsored by Boehringer Ingelheim Pharmaceuticals, Inc. (BIPI). Palli and Franchino-Elder are employees of BIPI. Frazer, DuCharme, Buikema, and Anderson are employees of Optum, which was contracted by BIPI to conduct this study. The authors received no direct compensation related to the development of the manuscript. BIPI was given the opportunity to review the manuscript for medical and scientific accuracy as well as intellectual property considerations.
Conflict of interest statement
This study was sponsored by Boehringer Ingelheim Pharmaceuticals, Inc. (BIPI). Palli and Franchino-Elder are employees of BIPI. Frazer, DuCharme, Buikema, and Anderson are employees of Optum, which was contracted by BIPI to conduct this study. The authors received no direct compensation related to the development of the manuscript. BIPI was given the opportunity to review the manuscript for medical and scientific accuracy as well as intellectual property considerations.
Figures

References
-
- Ford ES, Murphy LB, Khavjou O, Giles WH, Holt JB, Croft JB. Total and state-specific medical and absenteeism costs of COPD among adults aged ≥ 18 years in the United States for 2010 and projections through 2020. Chest. 2015;147(1):31-45. - PubMed
-
- Global Initiative for Chronic Obstructive Lung Disease, Inc. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease (2018 report). Available at: https://goldcopd.org/wp-content/uploads/2017/11/GOLD-2018-v6.0-FINAL-rev.... Accessed March 23, 2020.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

