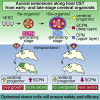
Axonal Extensions along Corticospinal Tracts from Transplanted Human Cerebral Organoids
- PMID: 32679062
- PMCID: PMC7419717
- DOI: 10.1016/j.stemcr.2020.06.016
Axonal Extensions along Corticospinal Tracts from Transplanted Human Cerebral Organoids
Abstract
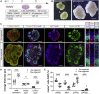
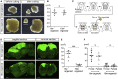
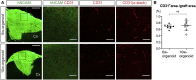
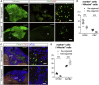
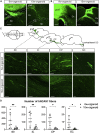
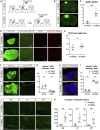
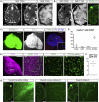
The reconstruction of lost neural circuits by cell replacement is a possible treatment for neurological deficits after cerebral cortex injury. Cerebral organoids can be a novel source for cell transplantation, but because the cellular composition of the organoids changes along the time course of the development, it remains unclear which developmental stage of the organoids is most suitable for reconstructing the corticospinal tract. Here, we transplanted human embryonic stem cell-derived cerebral organoids at 6 or 10 weeks after differentiation (6w- or 10w-organoids) into mouse cerebral cortices. 6w-organoids extended more axons along the corticospinal tract but caused graft overgrowth with a higher percentage of proliferative cells. Axonal extensions from 10w-organoids were smaller in number but were enhanced when the organoids were grafted 1 week after brain injury. Finally, 10w-organoids extended axons in cynomolgus monkey brains. These results contribute to the development of a cell-replacement therapy for brain injury and stroke.
Keywords: axonal extension; cell transplantation; cerebral cortex; cerebral organoid; corticospinal tract; developmental stage; graft overgrowth; human pluripotent stem cell; nonhuman primate; subcerebral projection neuron.
Copyright © 2020 The Authors. Published by Elsevier Inc. All rights reserved.
Figures
Similar articles
-
Viral delivery of L1CAM promotes axonal extensions by embryonic cerebral grafts in mouse brain.Stem Cell Reports. 2023 Apr 11;18(4):899-914. doi: 10.1016/j.stemcr.2023.02.012. Epub 2023 Mar 23. Stem Cell Reports. 2023. PMID: 36963389 Free PMC article.
-
Progranulin enhances the engraftment of transplanted human iPS cell-derived cerebral neurons.Stem Cells Transl Med. 2024 Nov 12;13(11):1113-1128. doi: 10.1093/stcltm/szae066. Stem Cells Transl Med. 2024. PMID: 39340829 Free PMC article.
-
Human neural stem cells promote corticospinal axons regeneration and synapse reformation in injured spinal cord of rats.Chin Med J (Engl). 2006 Aug 20;119(16):1331-8. Chin Med J (Engl). 2006. PMID: 16934177
-
Cerebral Cortex Generated from Pluripotent Stem Cells to Model Corticogenesis and Rebuild Cortical Circuits: In Vitro Veritas?Stem Cells Dev. 2019 Mar 15;28(6):361-369. doi: 10.1089/scd.2018.0233. Epub 2019 Feb 20. Stem Cells Dev. 2019. PMID: 30661489 Review.
-
Functional recovery through the plastic adaptation of organoid grafts in cortical tissue.Cell Mol Life Sci. 2025 Jun 9;82(1):227. doi: 10.1007/s00018-025-05767-w. Cell Mol Life Sci. 2025. PMID: 40490613 Free PMC article. Review.
Cited by
-
Human PSCs determine the competency of cerebral organoid differentiation via FGF signaling and epigenetic mechanisms.iScience. 2022 Sep 16;25(10):105140. doi: 10.1016/j.isci.2022.105140. eCollection 2022 Oct 21. iScience. 2022. PMID: 36185382 Free PMC article.
-
Towards human organ generation using interspecies blastocyst complementation: Challenges and perspectives for therapy.Front Cell Dev Biol. 2023 Jan 19;11:1070560. doi: 10.3389/fcell.2023.1070560. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 36743411 Free PMC article. Review.
-
Shedding light on latent pathogenesis and pathophysiology of mental disorders: The potential of iPS cell technology.Psychiatry Clin Neurosci. 2023 Jun;77(6):308-314. doi: 10.1111/pcn.13545. Epub 2023 Apr 4. Psychiatry Clin Neurosci. 2023. PMID: 36929185 Free PMC article. Review.
-
Midbrain organoids-development and applications in Parkinson's disease.Oxf Open Neurosci. 2023 Aug 18;2:kvad009. doi: 10.1093/oons/kvad009. eCollection 2023. Oxf Open Neurosci. 2023. PMID: 38596240 Free PMC article. Review.
-
A beginner's guide on the use of brain organoids for neuroscientists: a systematic review.Stem Cell Res Ther. 2023 Apr 15;14(1):87. doi: 10.1186/s13287-023-03302-x. Stem Cell Res Ther. 2023. PMID: 37061699 Free PMC article.
References
-
- Alcamo E.A., Chirivella L., Dautzenberg M., Dobreva G., Farinas I., Grosschedl R., McConnell S.K. Satb2 regulates callosal projection neuron identity in the developing cerebral cortex. Neuron. 2008;57:364–377. - PubMed
-
- Arlotta P., Molyneaux B.J., Chen J., Inoue J., Kominami R., Macklis J.D. Neuronal subtype-specific genes that control corticospinal motor neuron development in vivo. Neuron. 2005;45:207–221. - PubMed
-
- Britanova O., de Juan Romero C., Cheung A., Kwan K.Y., Schwark M., Gyorgy A., Vogel T., Akopov S., Mitkovski M., Agoston D. Satb2 is a postmitotic determinant for upper-layer neuron specification in the neocortex. Neuron. 2008;57:378–392. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

