Effect of Hydrophilic Polymers on Complexation Efficiency of Cyclodextrins in Enhancing Solubility and Release of Diflunisal
- PMID: 32679660
- PMCID: PMC7408593
- DOI: 10.3390/polym12071564
Effect of Hydrophilic Polymers on Complexation Efficiency of Cyclodextrins in Enhancing Solubility and Release of Diflunisal
Abstract
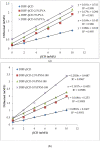
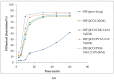
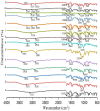
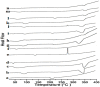
The effects of three hydrophilic polymers, namely, carboxymethyl cellulose sodium (CMC-Na), polyvinyl alcohol (PVA) and poloxamer-188 (PXM-188) on the solubility and dissolution of diflunisal (DIF) in complexation with β-cyclodextrin (βCD) or hydroxypropyl β-cyclodextrin (HPβCD), were investigated. The kneading method was used at different drug to cyclodextrin weight ratios. Increases in solubility and drug release were observed with the DIF/βCD and DIF/HPβCD complexes. The addition of hydrophilic polymers at 2.5, 5.0 and 10.0% w/w markedly improved the complexation and solubilizing efficiency of βCD and HPβCD. Fourier-transform infrared (FTIR) showed that DIF was successfully included into the cyclodextrin cavity. Differential scanning calorimetry (DSC) and X-ray diffractometry (XRD) confirmed stronger drug amorphization and entrapment in the molecular cage of cyclodextrins. The addition of PVA, CMC-Na or PXM-188 reduced further the intensity of the DIF endothermic peak. Most of the sharp and intense peaks of DIF disappeared with the addition of hydrophilic polymers. In conclusion, PXM-188 at a weight ratio of 10.0% w/w was the best candidate in enhancing the solubility, stability and release of DIF.
Keywords: complexation; diflunisal; dissolution rate; hydrophilic polymers; hydroxypropyl β-cyclodextrin; β-cyclodextrin.
Conflict of interest statement
There is no conflict of interest among the listed authors.
Figures



References
-
- Krishnaiah Y.S. Pharmaceutical technologies for enhancing oral bioavailability of poorly soluble drugs. J. Bioequiv. Bioavailab. 2010;2:28–36. doi: 10.4172/jbb.1000027. - DOI
-
- Kumar A., Sahoo S.K., Padhee K., Kochar P., Satapathy A., Pathak N. Review on solubility enhancement techniques for hydrophobic drugs. Pharm. Glob. 2011;3:1–7.
-
- Crupi V., Majolino D., Mele A., Rossi B., Trotta F., Venuti V. Modelling the interplay between covalent and physical interactions in cyclodextrin-based hydrogel: Effect of water confinement. Soft Matter. 2013;9:6457–6464. doi: 10.1039/c3sm50827g. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

