Pulmonary alveolar proteinosis: from classification to therapy
- PMID: 32684997
- PMCID: PMC7341616
- DOI: 10.1183/20734735.0018-2020
Pulmonary alveolar proteinosis: from classification to therapy
Abstract
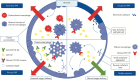
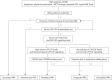
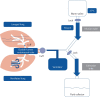
Pulmonary alveolar proteinosis (PAP) is a rare respiratory syndrome characterised by the accumulation of surfactant lipoproteins within the alveoli. According to various pathogenetic mechanisms and aetiologies, PAP is classified as primary, secondary or congenital. Primary PAP is led by a granulocyte-macrophage colony-stimulating factor (GM-CSF) signalling disruption; the autoimmune form is driven by the presence of anti GM-CSF autoantibodies and represents 90% of all the PAP cases; and the hereditary form is the result of mutations in genes encoding GM-CSF receptor. Secondary PAP is associated with various diseases causing a reduction in function and/or number of alveolar macrophages. Congenital PAP emerges as a consequence of corrupted surfactant production, due to mutations in surfactant proteins or lipid transporter, or mutations affecting lung development. The clinical manifestations are various, ranging from insidious onset to acute or progressive respiratory failure, including premature death within the first days of life in neonates with congenital surfactant production disorders. The diagnostic workup includes clinical and radiological assessment (respiratory function test, high-resolution chest computed tomography), laboratory tests (anti-GM-CSF autoantibodies dosage, GM-CSF serum level and GM-CSF signalling test), and genetic tests. Whole-lung lavage is the current gold standard of care of PAP; however, the therapeutic approach depends on the pathogenic form and disease severity, including GM-CSF augmentation strategies in autoimmune PAP and other promising new treatments.
Educational aims: To update knowledge about a rare respiratory syndrome, pulmonary alveolar proteinosis, in order to promote early diagnosis and correct management.To highlight recent treatment options based on pathogenesis and disease severity.
Copyright ©ERS 2020.
Conflict of interest statement
Conflict of interest: E. Salvaterra has nothing to disclose. Conflict of interest: I. Campo has nothing to disclose.
Figures

References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
