Redox and mTOR-dependent regulation of plasma lamellar calcium influx controls the senescence-associated secretory phenotype
- PMID: 32686475
- PMCID: PMC7787549
- DOI: 10.1177/1535370220943122
Redox and mTOR-dependent regulation of plasma lamellar calcium influx controls the senescence-associated secretory phenotype
Abstract
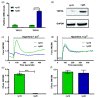
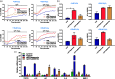
Through its ability to evoke responses from cells in a paracrine fashion, the senescence-associated secretory phenotype (SASP) has been linked to numerous age-associated disease pathologies including tumor invasion, cardiovascular dysfunction, neuroinflammation, osteoarthritis, and renal disease. Strategies which limit the amplitude and duration of SASP serve to delay age-related degenerative decline. Here we demonstrate that the SASP regulation is linked to shifts in intracellular Ca2+ homeostasis and strategies which rescue redox-dependent calcium entry including enzymatic H2O2 scavenging, TRP modulation, or mTOR inhibition block SASP and TRPC6 gene expression. As Ca2+ is indispensable for secretion from both secretory and non-secretory cells, it is exciting to speculate that the expression of plasma lamellar TRP channels critical for the maintenance of intracellular Ca2+ homeostasis may be coordinately regulated with the SASP.
Keywords: SASP; Senescence; TRPC6; calcium; hydrogen peroxide; mTOR.
Figures



Similar articles
-
Redox control of the senescence regulator interleukin-1α and the secretory phenotype.J Biol Chem. 2013 Nov 8;288(45):32149-32159. doi: 10.1074/jbc.M113.493841. Epub 2013 Sep 23. J Biol Chem. 2013. PMID: 24062309 Free PMC article.
-
mTOR regulates MAPKAPK2 translation to control the senescence-associated secretory phenotype.Nat Cell Biol. 2015 Sep;17(9):1205-17. doi: 10.1038/ncb3225. Epub 2015 Aug 17. Nat Cell Biol. 2015. PMID: 26280535 Free PMC article.
-
A senescence secretory switch mediated by PI3K/AKT/mTOR activation controls chemoprotective endothelial secretory responses.Genes Dev. 2016 Aug 15;30(16):1811-21. doi: 10.1101/gad.284851.116. Epub 2016 Aug 26. Genes Dev. 2016. PMID: 27566778 Free PMC article.
-
mTOR Activity and Autophagy in Senescent Cells, a Complex Partnership.Int J Mol Sci. 2021 Jul 29;22(15):8149. doi: 10.3390/ijms22158149. Int J Mol Sci. 2021. PMID: 34360912 Free PMC article. Review.
-
Rapamycin and the inhibition of the secretory phenotype.Exp Gerontol. 2017 Aug;94:89-92. doi: 10.1016/j.exger.2017.01.026. Epub 2017 Feb 4. Exp Gerontol. 2017. PMID: 28167236 Review.
Cited by
-
Mechanisms of aging, age-associated diseases, and lifespan determination.Exp Biol Med (Maywood). 2020 Nov;245(17):1529-1531. doi: 10.1177/1535370220955146. Epub 2020 Sep 9. Exp Biol Med (Maywood). 2020. PMID: 32903037 Free PMC article. No abstract available.
-
Therapy-Induced Cellular Senescence: Potentiating Tumor Elimination or Driving Cancer Resistance and Recurrence?Cells. 2024 Jul 30;13(15):1281. doi: 10.3390/cells13151281. Cells. 2024. PMID: 39120312 Free PMC article. Review.
-
Cardiac cell senescence: molecular mechanisms, key proteins and therapeutic targets.Cell Death Discov. 2024 Feb 14;10(1):78. doi: 10.1038/s41420-023-01792-5. Cell Death Discov. 2024. PMID: 38355681 Free PMC article. Review.
References
-
- Hayflick L, Moorhead PS. The serial cultivation of human diploid cell strains. Exp Cell Res 1961; 25:585–621 - PubMed
-
- Courtois-Cox S, Jones SL, Cichowski K. Many roads lead to oncogene-induced senescence. Oncogene 2008; 27:2801–9 - PubMed
-
- Wickens AP. Ageing and the free radical theory. Respir Physiol 2001; 128:379–91 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

