Human DECR1 is an androgen-repressed survival factor that regulates PUFA oxidation to protect prostate tumor cells from ferroptosis
- PMID: 32686647
- PMCID: PMC7386908
- DOI: 10.7554/eLife.54166
Human DECR1 is an androgen-repressed survival factor that regulates PUFA oxidation to protect prostate tumor cells from ferroptosis
Abstract
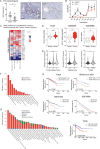
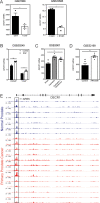
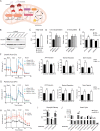
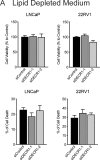
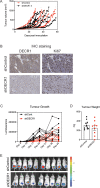
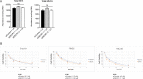
Fatty acid β-oxidation (FAO) is the main bioenergetic pathway in human prostate cancer (PCa) and a promising novel therapeutic vulnerability. Here we demonstrate therapeutic efficacy of targeting FAO in clinical prostate tumors cultured ex vivo, and identify DECR1, encoding the rate-limiting enzyme for oxidation of polyunsaturated fatty acids (PUFAs), as robustly overexpressed in PCa tissues and associated with shorter relapse-free survival. DECR1 is a negatively-regulated androgen receptor (AR) target gene and, therefore, may promote PCa cell survival and resistance to AR targeting therapeutics. DECR1 knockdown selectively inhibited β-oxidation of PUFAs, inhibited proliferation and migration of PCa cells, including treatment resistant lines, and suppressed tumor cell proliferation and metastasis in mouse xenograft models. Mechanistically, targeting of DECR1 caused cellular accumulation of PUFAs, enhanced mitochondrial oxidative stress and lipid peroxidation, and induced ferroptosis. These findings implicate PUFA oxidation via DECR1 as an unexplored facet of FAO that promotes survival of PCa cells.
Keywords: androgen; cancer biology; ferroptosis; human; lipid metabolism; mouse; prostate cancer.
© 2020, Nassar et al.
Conflict of interest statement
ZN, CM, JD, IB, SI, MC, MH, RS, MM, AD, JH, AS, LH, DL, LS, AH, JS, LB No competing interests declared
Figures








References
-
- Armstrong HK, Koay YC, Irani S, Das R, Nassar ZD, Selth LA, Centenera MM, McAlpine SR, Butler LM, Australian Prostate Cancer BioResource A novel class of Hsp90 C-Terminal modulators have Pre-Clinical efficacy in prostate tumor cells without induction of a heat shock response. The Prostate. 2016;76:1546–1559. doi: 10.1002/pros.23239. - DOI - PubMed
-
- Asangani IA, Dommeti VL, Wang X, Malik R, Cieslik M, Yang R, Escara-Wilke J, Wilder-Romans K, Dhanireddy S, Engelke C, Iyer MK, Jing X, Wu YM, Cao X, Qin ZS, Wang S, Feng FY, Chinnaiyan AM. Therapeutic targeting of BET bromodomain proteins in castration-resistant prostate Cancer. Nature. 2014;510:278–282. doi: 10.1038/nature13229. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
Grants and funding
- Early Career Fellowship,1138648/National Health and Medical Research Council/International
- Project Grants C16/15/073 and C32/17/052/KU Leuven/International
- Future Fellowship,FT130101004/Australian Research Council/International
- Beat Cancer Fellowship,PRF1117/Cancer Council South Australia/International
- Revolutionary Team Award,MRTA3/Movember Foundation/International
- Project Grant,1121057/National Health and Medical Research Council/International
- Project Grant,1100626/National Health and Medical Research Council/International
- Fellowship,1084178/National Health and Medical Research Council/International
- Young Investigator Award,YI 1417/Prostate Cancer Foundation of Australia/International
- Project Grant,1164798/Cure Cancer Australia Foundation/International
- Group Leader Award/EMBL Australia/International
- Robinson Fellowship/University of Sydney/International
- Project Grants G.0841.15 and G.0C22.19N/Fonds Wetenschappelijk Onderzoek/International
- 1138648/National Health and Medical Research Council/International
- 1121057/National Health and Medical Research Council/International
- 1100626/National Health and Medical Research Council/International
- 1084178/National Health and Medical Research Council/International
- YI 1417/Prostate Cancer Foundation of Australia/International
- 1164798/Cure Cancer Australia Foundation/International
- FT130101004/Australian Research Council/International
- PRF1117/Cancer Council South Australia/International
- MRTA3/Movember Foundation/International
LinkOut - more resources
Full Text Sources
Medical
Research Materials

