Wnt signaling and Loxl2 promote aggressive osteosarcoma
- PMID: 32686768
- PMCID: PMC7608146
- DOI: 10.1038/s41422-020-0370-1
Wnt signaling and Loxl2 promote aggressive osteosarcoma
Abstract
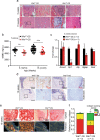
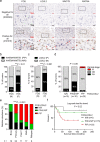
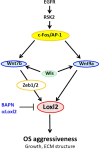
Osteosarcoma (OS) is the most frequent primary malignant bone tumor in urgent need of better therapies. Using genetically modified mouse models (GEMMs), we demonstrate that Wnt signaling promotes c-Fos-induced OS formation via the actions of the collagen-modifying enzyme Loxl2. c-Fos/AP-1 directly regulates the expression of the Wnt ligands Wnt7b and Wnt9a in OS cells through promoter binding, and Wnt7b and Wnt9a in turn promote Loxl2 expression in murine and human OS cells through the transcription factors Zeb1 and Zeb2. Concordantly, inhibition of Wnt ligand secretion by inactivating the Wnt-less (Wls) gene in osteoblasts in c-Fos GEMMs either early or in a therapeutic setting reduces Loxl2 expression and progression of OS. Wls-deficient osteosarcomas proliferate less, are less mineralized and are enriched in fibroblastic cells surrounded by collagen fibers. Importantly, Loxl2 inhibition using either the pan-Lox inhibitor BAPN or a specific inducible shRNA reduces OS cell proliferation in vitro and decreases tumor growth and lung colonization in murine and human orthotopic OS transplantation models. Finally, OS development is delayed in c-Fos GEMMs treated with BAPN or with specific Loxl2 blocking antibodies. Congruently, a strong correlation between c-FOS, LOXL2 and WNT7B/WNT9A expression is observed in human OS samples, and c-FOS/LOXL2 co-expression correlates with OS aggressiveness and decreased patient survival. Therefore, therapeutic targeting of Wnt and/or Loxl2 should be considered to potentiate the inadequate current treatments for pediatric, recurrent, and metastatic OS.
Conflict of interest statement
A.M.V. is an employee of Gilead Sciences. The remaining authors declare no competing interests.
Figures





Similar articles
-
miR-342-5p inhibits osteosarcoma cell growth, migration, invasion, and sensitivity to Doxorubicin through targeting Wnt7b.Cell Cycle. 2019 Dec;18(23):3325-3336. doi: 10.1080/15384101.2019.1676087. Epub 2019 Oct 10. Cell Cycle. 2019. PMID: 31601147 Free PMC article.
-
NKD2, a negative regulator of Wnt signaling, suppresses tumor growth and metastasis in osteosarcoma.Oncogene. 2015 Sep 24;34(39):5069-79. doi: 10.1038/onc.2014.429. Epub 2015 Jan 12. Oncogene. 2015. PMID: 25579177 Free PMC article.
-
Tumor-secreted LOXL2 activates fibroblasts through FAK signaling.Mol Cancer Res. 2013 Nov;11(11):1425-36. doi: 10.1158/1541-7786.MCR-13-0033-T. Epub 2013 Sep 5. Mol Cancer Res. 2013. PMID: 24008674 Free PMC article.
-
High Expression of XRCC6 Promotes Human Osteosarcoma Cell Proliferation through the β-Catenin/Wnt Signaling Pathway and Is Associated with Poor Prognosis.Int J Mol Sci. 2016 Jul 22;17(7):1188. doi: 10.3390/ijms17071188. Int J Mol Sci. 2016. PMID: 27455247 Free PMC article.
-
Wnt signaling in osteosarcoma.Adv Exp Med Biol. 2014;804:33-45. doi: 10.1007/978-3-319-04843-7_2. Adv Exp Med Biol. 2014. PMID: 24924167 Review.
Cited by
-
The role of Evi/Wntless in exporting Wnt proteins.Development. 2023 Feb 15;150(3):dev201352. doi: 10.1242/dev.201352. Epub 2023 Feb 10. Development. 2023. PMID: 36763105 Free PMC article. Review.
-
Nerve growth factor promotes lysyl oxidase-dependent chondrosarcoma cell metastasis by suppressing miR-149-5p synthesis.Cell Death Dis. 2021 Nov 23;12(12):1101. doi: 10.1038/s41419-021-04392-2. Cell Death Dis. 2021. PMID: 34815382 Free PMC article.
-
CGREF1 modulates osteosarcoma proliferation by regulating the cell cycle through the Wnt/β-catenin signaling pathway.Mol Med. 2024 Dec 20;30(1):260. doi: 10.1186/s10020-024-01038-9. Mol Med. 2024. PMID: 39707194 Free PMC article.
-
Inhibition of dopamine receptor D1 signaling promotes human bile duct cancer progression via WNT signaling.Cancer Sci. 2023 Apr;114(4):1324-1336. doi: 10.1111/cas.15676. Epub 2022 Dec 14. Cancer Sci. 2023. PMID: 36441110 Free PMC article.
-
Polypeptide N-Acetylgalactosaminyltransferase 14 (GALNT14) as a Chemosensitivity-Related Biomarker for Osteosarcoma.J Oncol. 2023 Feb 2;2023:1083423. doi: 10.1155/2023/1083423. eCollection 2023. J Oncol. 2023. PMID: 38024474 Free PMC article.
References
-
- Heaney RP, et al. Peak bone mass. Osteoporos. Int. 2000;11:985–1009. - PubMed
-
- Fernandes DA, Costa AA, Lahdenperä P. Osteosarcoma genetics and epigenetics: emerging biology and candidate therapies. Int. J. Constr. Manag. 2018;18:482–496.
-
- Link MP, et al. The effect of adjuvant chemotherapy on relapse-free survival in patients with osteosarcoma of the extremity. N. Engl. J. Med. 1986;314:1600–1606. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

