Changes in ferrous iron and glutathione promote ferroptosis and frailty in aging Caenorhabditis elegans
- PMID: 32690135
- PMCID: PMC7373428
- DOI: 10.7554/eLife.56580
Changes in ferrous iron and glutathione promote ferroptosis and frailty in aging Caenorhabditis elegans
Abstract
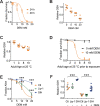
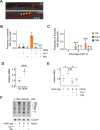
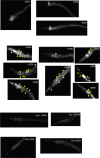
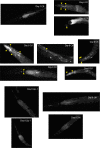
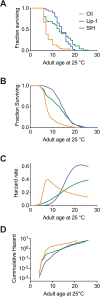
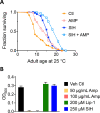
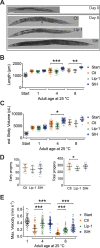
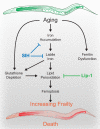
All eukaryotes require iron. Replication, detoxification, and a cancer-protective form of regulated cell death termed ferroptosis, all depend on iron metabolism. Ferrous iron accumulates over adult lifetime in Caenorhabditis elegans. Here, we show that glutathione depletion is coupled to ferrous iron elevation in these animals, and that both occur in late life to prime cells for ferroptosis. We demonstrate that blocking ferroptosis, either by inhibition of lipid peroxidation or by limiting iron retention, mitigates age-related cell death and markedly increases lifespan and healthspan. Temporal scaling of lifespan is not evident when ferroptosis is inhibited, consistent with this cell death process acting at specific life phases to induce organismal frailty, rather than contributing to a constant aging rate. Because excess age-related iron elevation in somatic tissue, particularly in brain, is thought to contribute to degenerative disease, post-developmental interventions to limit ferroptosis may promote healthy aging.
Keywords: C. elegans; biochemistry; chemical biology; ferroptosis; fitness; frailty; glutathione; iron; lifespan.
© 2020, Jenkins et al.
Conflict of interest statement
NJ, SJ, AS, FS, TS, MC, DR No competing interests declared, AB AIB is a paid consultant for, and has a profit share interest in, Collaborative Medicinal Development Pty Ltd and is an inventor on patent application 15/505,384, 2017, which covers the method of reducing senescence in a mammal by reducing the concentration of non-ferritin iron, GM GM is an inventor on patent application 15/505,384, 2017, which covers the method of reducing senescence in a mammal by reducing the concentration of non-ferritin iron
Figures



Similar articles
-
Contribution of Ferroptosis to Aging and Frailty.Rejuvenation Res. 2020 Oct;23(5):434-438. doi: 10.1089/rej.2020.2390. Rejuvenation Res. 2020. PMID: 32977738
-
Inhibition of Ferroptosis Delays Aging and Extends Healthspan Across Multiple Species.Adv Sci (Weinh). 2025 Jun;12(22):e2416559. doi: 10.1002/advs.202416559. Epub 2025 Mar 31. Adv Sci (Weinh). 2025. PMID: 40162524 Free PMC article.
-
Isobavachalcone confers protection against Cryptococcus neoformans-induced ferroptosis in Caenorhabditis elegans via lifespan extension and GSH-GPX-1 axis modulation.J Hazard Mater. 2025 Jul 15;492:137969. doi: 10.1016/j.jhazmat.2025.137969. Epub 2025 Mar 18. J Hazard Mater. 2025. PMID: 40154123
-
Ferroptosis: Role of lipid peroxidation, iron and ferritinophagy.Biochim Biophys Acta Gen Subj. 2017 Aug;1861(8):1893-1900. doi: 10.1016/j.bbagen.2017.05.019. Epub 2017 May 24. Biochim Biophys Acta Gen Subj. 2017. PMID: 28552631 Review.
-
Mechanisms and regulations of ferroptosis.Front Immunol. 2023 Oct 6;14:1269451. doi: 10.3389/fimmu.2023.1269451. eCollection 2023. Front Immunol. 2023. PMID: 37868994 Free PMC article. Review.
Cited by
-
The Impairment of Endothelial Autophagy Accelerates Renal Senescence by Ferroptosis and NLRP3 Inflammasome Signaling Pathways with the Disruption of Endothelial Barrier.Antioxidants (Basel). 2024 Jul 23;13(8):886. doi: 10.3390/antiox13080886. Antioxidants (Basel). 2024. PMID: 39199133 Free PMC article.
-
Glutathione for Food and Health Applications with Emphasis on Extraction, Identification, and Quantification Methods: A Review.Metabolites. 2023 Mar 24;13(4):465. doi: 10.3390/metabo13040465. Metabolites. 2023. PMID: 37110125 Free PMC article. Review.
-
Functional Equivalence of Insulin and IGF-1 in the In Vitro Culture of Chicken Primordial Germ Cells.Genes (Basel). 2025 Apr 24;16(5):481. doi: 10.3390/genes16050481. Genes (Basel). 2025. PMID: 40428303 Free PMC article.
-
Unveiling ferroptosis: a new frontier in skin disease research.Front Immunol. 2024 Oct 4;15:1485523. doi: 10.3389/fimmu.2024.1485523. eCollection 2024. Front Immunol. 2024. PMID: 39430757 Free PMC article. Review.
-
Platelet-derived exosomes alleviate tendon stem/progenitor cell senescence and ferroptosis by regulating AMPK/Nrf2/GPX4 signaling and improve tendon-bone junction regeneration in rats.J Orthop Surg Res. 2024 Jun 28;19(1):382. doi: 10.1186/s13018-024-04869-8. J Orthop Surg Res. 2024. PMID: 38943181 Free PMC article.
References
-
- Berry AJ, O’Neill HSC, Jayasuriya KD, Campbell SJ, Foran GJ. XANES calibrations for the oxidation state of iron in a silicate glass. American Mineralogist. 2003;88:967–977. doi: 10.2138/am-2003-0704. - DOI
Publication types
MeSH terms
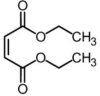
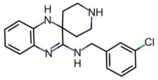
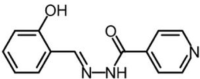
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

