Depolarization-Dependent C-Raf Signaling Promotes Hyperexcitability and Reduces Opioid Sensitivity of Isolated Nociceptors after Spinal Cord Injury
- PMID: 32690613
- PMCID: PMC7486665
- DOI: 10.1523/JNEUROSCI.0810-20.2020
Depolarization-Dependent C-Raf Signaling Promotes Hyperexcitability and Reduces Opioid Sensitivity of Isolated Nociceptors after Spinal Cord Injury
Abstract
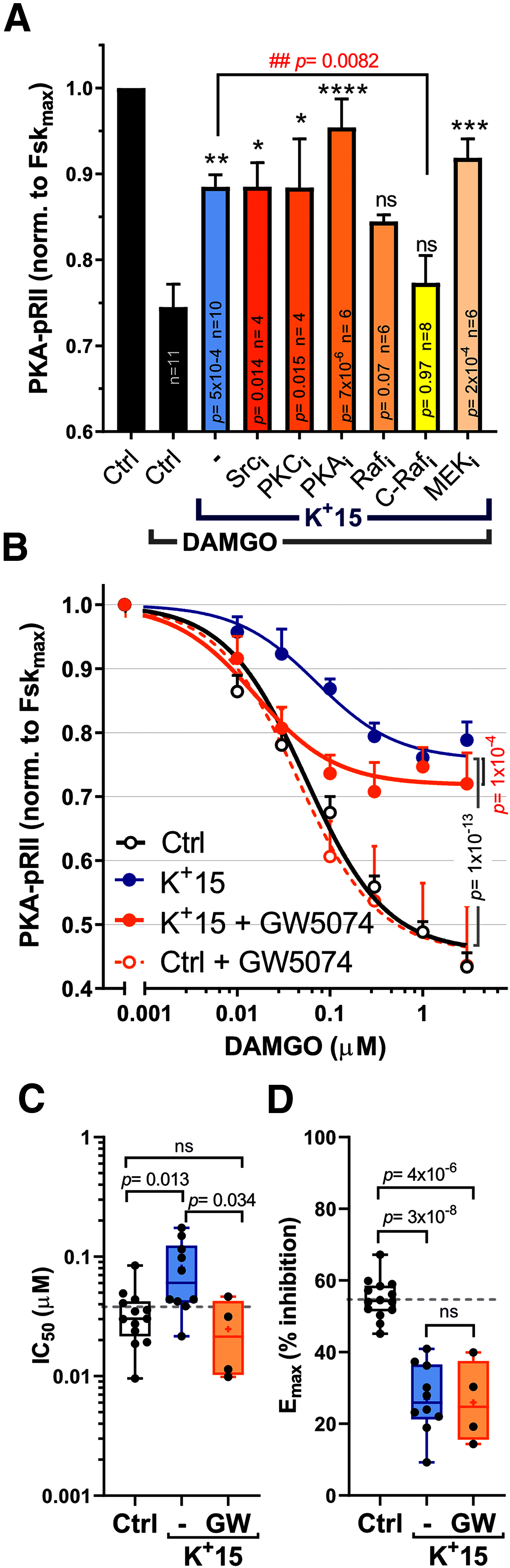
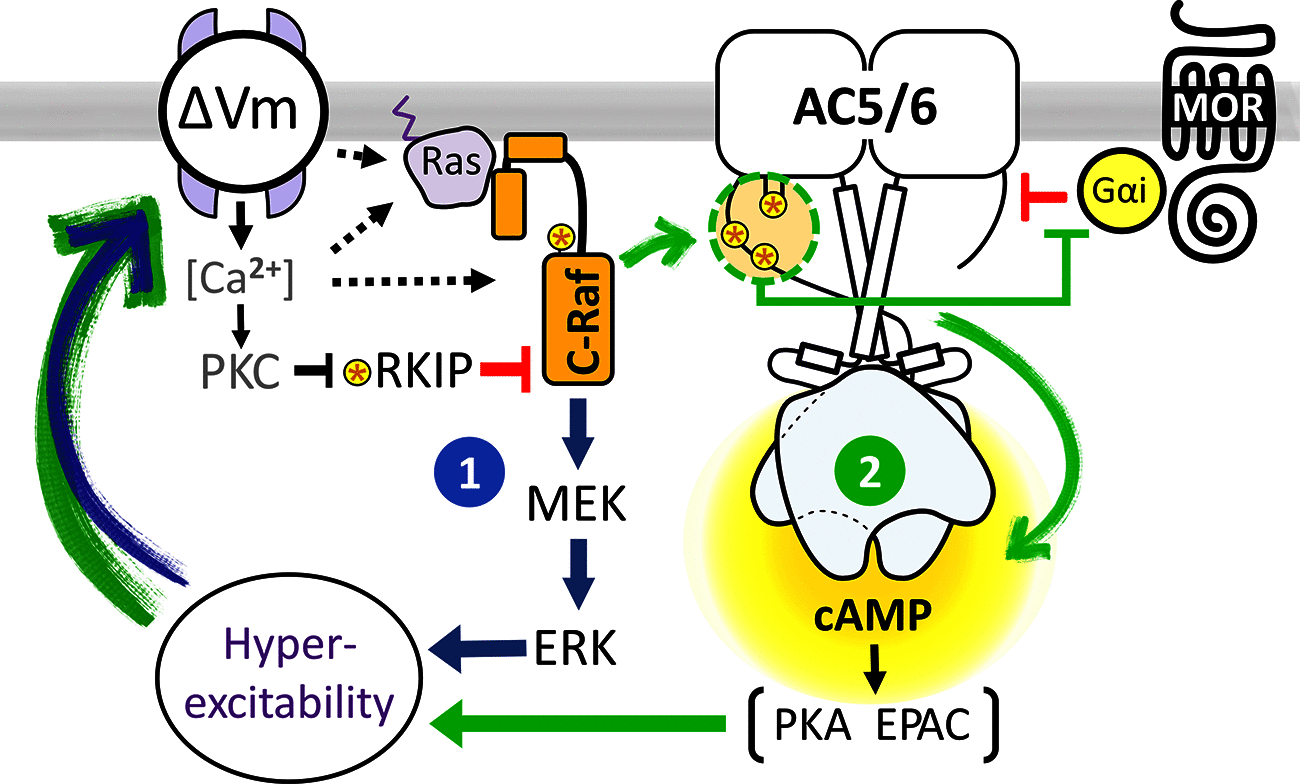
Chronic pain caused by spinal cord injury (SCI) is notoriously resistant to treatment, particularly by opioids. After SCI, DRG neurons show hyperactivity and chronic depolarization of resting membrane potential (RMP) that is maintained by cAMP signaling through PKA and EPAC. Importantly, SCI also reduces the negative regulation by Gαi of adenylyl cyclase and its production of cAMP, independent of alterations in G protein-coupled receptors and/or G proteins. Opioid reduction of pain depends on coupling of opioid receptors to Gαi/o family members. Combining high-content imaging and cluster analysis, we show that in male rats SCI decreases opioid responsiveness in vitro within a specific subset of small-diameter nociceptors that bind isolectin B4. This SCI effect is mimicked in nociceptors from naive animals by a modest 5 min depolarization of RMP (15 mm K+; -45 mV), reducing inhibition of cAMP signaling by μ-opioid receptor agonists DAMGO and morphine. Disinhibition and activation of C-Raf by depolarization-dependent phosphorylation are central to these effects. Expression of an activated C-Raf reduces sensitivity of adenylyl cyclase to opioids in nonexcitable HEK293 cells, whereas inhibition of C-Raf or treatment with the hyperpolarizing drug retigabine restores opioid responsiveness and blocks spontaneous activity of nociceptors after SCI. Inhibition of ERK downstream of C-Raf also blocks SCI-induced hyperexcitability and depolarization, without direct effects on opioid responsiveness. Thus, depolarization-dependent C-Raf and downstream ERK activity maintain a depolarized RMP and nociceptor hyperactivity after SCI, providing a self-reinforcing mechanism to persistently promote nociceptor hyperexcitability and limit the therapeutic effectiveness of opioids.SIGNIFICANCE STATEMENT Chronic pain induced by spinal cord injury (SCI) is often permanent and debilitating, and usually refractory to treatment with analgesics, including opioids. SCI-induced pain in a rat model has been shown to depend on persistent hyperactivity in primary nociceptors (injury-detecting sensory neurons), associated with a decrease in the sensitivity of adenylyl cyclase production of cAMP to inhibitory Gαi proteins in DRGs. This study shows that SCI and one consequence of SCI (chronic depolarization of resting membrane potential) decrease sensitivity to opioid-mediated inhibition of cAMP and promote hyperactivity of nociceptors by enhancing C-Raf activity. ERK activation downstream of C-Raf is necessary for maintaining ongoing depolarization and hyperactivity, demonstrating an unexpected positive feedback loop to persistently promote pain.
Keywords: MAP kinase; adenylyl cyclase; chronic pain; nociceptive; opiate; resting membrane potential.
Copyright © 2020 the authors.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
