Ex vivo mesoscopic diffusion MRI correlates with seizure frequency in patients with uncontrolled mesial temporal lobe epilepsy
- PMID: 32691978
- PMCID: PMC7555080
- DOI: 10.1002/hbm.25139
Ex vivo mesoscopic diffusion MRI correlates with seizure frequency in patients with uncontrolled mesial temporal lobe epilepsy
Abstract
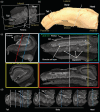
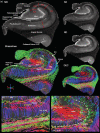
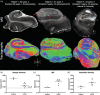
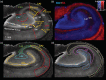
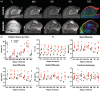
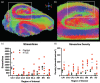
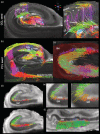
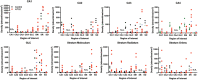
The role of hippocampal connectivity in mesial temporal lobe epilepsy (mTLE) remains poorly understood. The use of ex vivo hippocampal samples excised from patients with mTLE affords mesoscale diffusion magnetic resonance imaging (MRI) to identify individual cell layers, such as the pyramidal (PCL) and granule cell layers (GCL), which are thought to be impacted by seizure activity. Diffusion tensor imaging (DTI) of control (n = 3) and mTLE (n = 7) hippocampi on an 11.7 T MRI scanner allowed us to reveal intra-hippocampal connectivity and evaluate how epilepsy affected mean (MD), axial (AD), and radial diffusivity (RD), as well as fractional anisotropy (FA). Regional measurements indicated a volume loss in the PCL of the cornu ammonis (CA) 1 subfield in mTLE patients compared to controls, which provided anatomical context. Diffusion measurements, as well as streamline density, were generally higher in mTLE patients compared to controls, potentially reflecting differences due to tissue fixation. mTLE measurements were more variable than controls. This variability was associated with disease severity, as indicated by a strong correlation (r = 0.87) between FA in the stratum radiatum and the frequency of seizures in patients. MD and RD of the PCL in subfields CA3 and CA4 also correlated strongly with disease severity. No correlation of MR measures with disease duration was evident. These results reveal the potential of mesoscale diffusion MRI to examine layer-specific diffusion changes and connectivity to determine how these relate to clinical measures. Improving the visualization of intra-hippocampal connectivity will advance the development of novel hypotheses about seizure networks.
Keywords: biomarker; connectivity; diffusion mri; epilepsy; hippocampus; mesoscale; surgical resection; tractography.
© 2020 The Authors. Human Brain Mapping published by Wiley Periodicals LLC.
Conflict of interest statement
The authors have no personal financial or institutional interest in the results described in this article.
Figures
Similar articles
-
Mesoscale diffusion magnetic resonance imaging of the ex vivo human hippocampus.Hum Brain Mapp. 2020 Oct 15;41(15):4200-4218. doi: 10.1002/hbm.25119. Epub 2020 Jul 4. Hum Brain Mapp. 2020. PMID: 32621364 Free PMC article.
-
Hemispheric Regional Based Analysis of Diffusion Tensor Imaging and Diffusion Tensor Tractography in Patients with Temporal Lobe Epilepsy and Correlation with Patient outcomes.Sci Rep. 2019 Jan 18;9(1):215. doi: 10.1038/s41598-018-36818-x. Sci Rep. 2019. PMID: 30659215 Free PMC article.
-
Alterations in the hippocampal-thalamic pathway underlying secondarily generalized tonic-clonic seizures in mesial temporal lobe epilepsy: A diffusion tensor imaging study.Epilepsia. 2019 Jan;60(1):121-130. doi: 10.1111/epi.14614. Epub 2018 Nov 26. Epilepsia. 2019. PMID: 30478929
-
Hippocampal sclerosis--origins and imaging.Epilepsia. 2012 Sep;53 Suppl 4:19-33. doi: 10.1111/j.1528-1167.2012.03610.x. Epilepsia. 2012. PMID: 22946718 Review.
-
Temporal lobe epilepsy: where do the seizures really begin?Epilepsy Behav. 2009 Jan;14 Suppl 1(Suppl 1):32-7. doi: 10.1016/j.yebeh.2008.09.017. Epub 2008 Oct 31. Epilepsy Behav. 2009. PMID: 18848643 Free PMC article. Review.
Cited by
-
Mapping mesoscale connectivity within the human hippocampus.Neuroimage. 2023 Nov 15;282:120406. doi: 10.1016/j.neuroimage.2023.120406. Epub 2023 Oct 11. Neuroimage. 2023. PMID: 37827206 Free PMC article.
-
Ex vivo 100 μm isotropic diffusion MRI-based tractography of connectivity changes in the end-stage R6/2 mouse model of Huntington's disease.Neuroprotection. 2023 Sep;1(1):66-83. doi: 10.1002/nep3.14. Epub 2022 Dec 20. Neuroprotection. 2023. PMID: 37745674 Free PMC article.
-
Acquisition and Analysis of Excised Neocortex from Pediatric Patients with Focal Cortical Dysplasia Using Mesoscale Diffusion MRI.Diagnostics (Basel). 2023 Apr 24;13(9):1529. doi: 10.3390/diagnostics13091529. Diagnostics (Basel). 2023. PMID: 37174921 Free PMC article.
-
Layer-specific microstructural patterns of anterior hippocampus in Alzheimer's disease with ex vivo diffusion MRI at 14.1 T.Hum Brain Mapp. 2023 Feb 1;44(2):458-471. doi: 10.1002/hbm.26062. Epub 2022 Sep 2. Hum Brain Mapp. 2023. PMID: 36053237 Free PMC article.
-
3D MERMAID: 3D Multi-shot enhanced recovery motion artifact insensitive diffusion for submillimeter, multi-shell, and SNR-efficient diffusion imaging.Magn Reson Med. 2025 Jun;93(6):2311-2330. doi: 10.1002/mrm.30436. Epub 2025 Mar 4. Magn Reson Med. 2025. PMID: 40035173 Free PMC article.
References
-
- Adler, D. H. , Wisse, L. E. M. , Ittyerah, R. , Pluta, J. B. , Ding, S. L. , Xie, L. , … Yushkevich, P. A. (2018). Characterizing the human hippocampus in aging and Alzheimer's disease using a computational atlas derived from ex vivo MRI and histology. Proceedings of the National Academy of Sciences of the United States of America, 115(16), 4252–4257. 10.1073/pnas.1801093115 - DOI - PMC - PubMed
-
- Alizadeh, M. , Kozlowski, L. , Muller, J. , Ashraf, N. , Shahrampour, S. , Mohamed, F. B. , … Sharan, A. (2019). Hemispheric regional based analysis of diffusion tensor imaging and diffusion tensor tractozgraphy in patients with temporal lobe epilepsy and correlation with patient outcomes. Scientific Reports, 9(1), 215 10.1038/s41598-018-36818-x - DOI - PMC - PubMed
-
- Bao, Y. , He, R. , Zeng, Q. , Zhu, P. , Zheng, R. , & Xu, H. (2018). Investigation of microstructural abnormalities in white and gray matter around hippocampus with diffusion tensor imaging (DTI) in temporal lobe epilepsy (TLE). Epilepsy & Behavior, 83, 44–49. 10.1016/j.yebeh.2017.12.002 - DOI - PubMed
-
- Bartsch, T. (2012). The clinical neurobiology of the hippocampus: An integrative view. Oxford, UK: Oxford University Press.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

