Elevated Liver Biochemistries in Hospitalized Chinese Patients With Severe COVID-19: Systematic Review and Meta-analysis
- PMID: 32692464
- PMCID: PMC7405102
- DOI: 10.1002/hep.31472
Elevated Liver Biochemistries in Hospitalized Chinese Patients With Severe COVID-19: Systematic Review and Meta-analysis
Abstract
Background and aims: Several recent studies have reported an abnormal liver chemistry profile among patients with coronavirus disease 2019 (COVID-19), although its clinical significance remains unknown.
Approach and results: This systematic review and meta-analysis identified six studies of 586 patients delineating liver chemistries among patients with severe/critical illness versus mild cases of COVID-19 infection. Patients with severe/critical illness with COVID-19 infection have increased prevalence of coronary artery disease, cerebrovascular disease, and chronic obstructive pulmonary disease as compared with mild cases. A significant association between severe/critical COVID-19 infections with elevations in aspartate aminotransferase (pooled mean difference [MD], 11.70 U/L; 95% confidence interval [CI], 2.97, 20.43; P = 0.009), elevated total bilirubin (pooled MD, 0.14 mg/dL; 95% CI, 0.06, 0.22; P = 0.0005), and decreased albumin (pooled MD, -0.68 g/L; 95% CI, -0.81, -0.55; P < 0.00001) was noted. There was also a trend toward elevated alanine aminotransferase levels among these severe cases (pooled MD, 8.84 U/L; 95% CI, -2.28, 19.97; P = 0.12); however, this did not reach statistical significance. More severe/critically ill cases were associated with leukocytosis, neutrophilia, lymphopenia, elevated creatinine kinase, elevated lactate dehydrogenase (LDH), and elevated prothrombin time (PT).
Conclusions: Comorbidities, including coronary artery disease, cerebrovascular disease and chronic obstructive pulmonary disease, are more prevalent in hospitalized Chinese patients with severe/critical illness from COVID-19, and these patients are more likely to manifest with abnormal liver chemistries. Further prospective studies are crucial to understand the pathophysiologic mechanisms underlying the hepatic manifestations of the novel COVID-19 infection and its clinical significance.
© 2020 by the American Association for the Study of Liver Diseases.
Figures



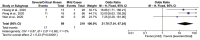
Comment in
-
Letter to the Editor: More Efforts for Detecting the Elevated Liver Biochemistries in Patients With Severe COVID-19.Hepatology. 2021 Nov;74(5):2903. doi: 10.1002/hep.31632. Hepatology. 2021. PMID: 33188700 Free PMC article. No abstract available.
References
-
- Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group . Preferred reporting items for systematic reviews and meta‐analyses: the PRISMA statement. Ann Intern Med 2009;151:264‐269, w64. - PubMed
-
- Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, et al. Meta‐analysis of observational studies in epidemiology: a proposal for reporting. Meta‐analysis of Observational Studies in Epidemiology (MOOSE) group. JAMA 2000;283:2008‐2012. - PubMed
-
- Wells G, Shea B, O’Connell D, Peterson J, Welch V, Losos M, et al. The Newcastle‐Ottawa Scale [NOS] for assessing the quality of nonrandomised studies in meta‐analyses. 2013. http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. Accessed March 30, 2020.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

