Lactate: the ugly duckling of energy metabolism
- PMID: 32694798
- PMCID: PMC7983055
- DOI: 10.1038/s42255-020-0243-4
Lactate: the ugly duckling of energy metabolism
Abstract
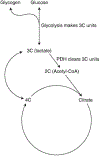
Lactate, perhaps the best-known metabolic waste product, was first isolated from sour milk, in which it is produced by lactobacilli. Whereas microbes also generate other fermentation products, such as ethanol or acetone, lactate dominates in mammals. Lactate production increases when the demand for ATP and oxygen exceeds supply, as occurs during intense exercise and ischaemia. The build-up of lactate in stressed muscle and ischaemic tissues has established lactate's reputation as a deleterious waste product. In this Perspective, we summarize emerging evidence that, in mammals, lactate also serves as a major circulating carbohydrate fuel. By providing mammalian cells with both a convenient source and sink for three-carbon compounds, circulating lactate enables the uncoupling of carbohydrate-driven mitochondrial energy generation from glycolysis. Lactate and pyruvate together serve as a circulating redox buffer that equilibrates the NADH/NAD ratio across cells and tissues. This reconceptualization of lactate as a fuel-analogous to how Hans Christian Andersen's ugly duckling is actually a beautiful swan-has the potential to reshape the field of energy metabolism.
Figures
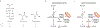
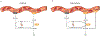
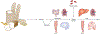
References
-
- Barron JT, Gu L & Parrillo JE Malate-aspartate shuttle, cytoplasmic NADH redox potential, and energetics in vascular smooth muscle. J. Mol. Cell. Cardiol 30, 1571–1579 (1998). - PubMed
-
- LaNoue KF & Williamson JR Interrelationships between malate-aspartate shuttle and citric acid cycle in rat heart mitochondria. Metabolism 20, 119–140 (1971). - PubMed
-
- Houstĕk J, Cannon B & Lindberg O Gylcerol-3-phosphate shuttle and its function in intermediary metabolism of hamster brown-adipose tissue. Eur. J. Biochem 54, 11–18 (1975). - PubMed
-
- Jorfeldt L, Juhlin-Dannfelt A & Karlsson J Lactate release in relation to tissue lactate in human skeletal muscle during exercise. J. Appl. Physiol 44, 350–352 (1978). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

