An Approach towards a GMP Compliant In-Vitro Expansion of Human Adipose Stem Cells for Autologous Therapies
- PMID: 32698363
- PMCID: PMC7552624
- DOI: 10.3390/bioengineering7030077
An Approach towards a GMP Compliant In-Vitro Expansion of Human Adipose Stem Cells for Autologous Therapies
Abstract
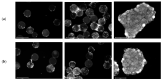
Human Adipose Tissue Stem Cells (hASCs) are a valuable source of cells for clinical applications (e.g., treatment of acute myocardial infarction and inflammatory diseases), especially in the field of regenerative medicine. However, for autologous (patient-specific) and allogeneic (off-the-shelf) hASC-based therapies, in-vitro expansion is necessary prior to the clinical application in order to achieve the required cell numbers. Safe, reproducible and economic in-vitro expansion of hASCs for autologous therapies is more problematic because the cell material changes for each treatment. Moreover, cell material is normally isolated from non-healthy or older patients, which further complicates successful in-vitro expansion. Hence, the goal of this study was to perform cell expansion studies with hASCs isolated from two different patients/donors (i.e., different ages and health statuses) under xeno- and serum-free conditions in static, planar (2D) and dynamically mixed (3D) cultivation systems. Our primary aim was I) to compare donor variability under in-vitro conditions and II) to develop and establish an unstructured, segregated growth model as a proof-of-concept study. Maximum cell densities of between 0.49 and 0.65 × 105 hASCs/cm2 were achieved for both donors in 2D and 3D cultivation systems. Cell growth under static and dynamically mixed conditions was comparable, which demonstrated that hydrodynamic stresses (P/V = 0.63 W/m3, τnt = 4.96 × 10-3 Pa) acting at Ns1u (49 rpm for 10 g/L) did not negatively affect cell growth, even under serum-free conditions. However, donor-dependent differences in the cell size were found, which resulted in significantly different maximum cell densities for each of the two donors. In both cases, stemness was well maintained under static 2D and dynamic 3D conditions, as long as the cells were not hyperconfluent. The optimal point for cell harvesting was identified as between cell densities of 0.41 and 0.56 × 105 hASCs/cm2 (end of exponential growth phase). The growth model delivered reliable predictions for cell growth, substrate consumption and metabolite production in both types of cultivation systems. Therefore, the model can be used as a basis for future investigations in order to develop a robust MC-based hASC production process for autologous therapies.
Keywords: UrSuppe stem cell culture medium; autologous therapy; human adipose stem cells (hASCs); kinetic growth modeling; segregated and unstructured growth model; serum- and xeno-free conditions.
Conflict of interest statement
The authors declare that there is no conflict of interests regarding the publication of this paper.
Figures








References
-
- De Francesco F., Mannucci S., Conti G., Prè E.D., Sbarbati A., Riccio M. A non-enzymatic method to obtain a fat tissue derivative highly enriched in adipose stem cells (ASCs) from human lipoaspirates: Preliminary results. Int. J. Mol. Sci. 2018;19:2061. doi: 10.3390/ijms19072061. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

