A novel coordination complex of platinum (PT) induces cell death in colorectal cancer by altering redox balance and modulating MAPK pathway
- PMID: 32703189
- PMCID: PMC7376665
- DOI: 10.1186/s12885-020-07165-w
A novel coordination complex of platinum (PT) induces cell death in colorectal cancer by altering redox balance and modulating MAPK pathway
Erratum in
-
Correction to: A novel coordination complex of platinum (PT) induces cell death in colorectal cancer by altering redox balance and modulating MAPK pathway.BMC Cancer. 2020 Sep 1;20(1):834. doi: 10.1186/s12885-020-07245-x. BMC Cancer. 2020. PMID: 32873240 Free PMC article.
Abstract
Background: Colorectal cancer (CRC) is a heterogeneous tumor having various genetic alterations. The current treatment options had limited impact on disease free survival due to therapeutic resistance. Novel anticancer agents are needed to treat CRC specifically metastatic colorectal cancer. A novel coordination complex of platinum, (salicylaldiminato)Pt(II) complex with dimethylpropylene linkage (PT) exhibited potential anti-cancer activity. In this study, we explored the molecular mechanism of PT-induced cell death in colorectal cancer.
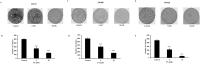
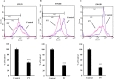
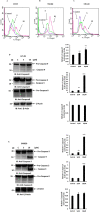
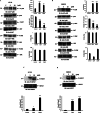
Methods: Colony formation was evaluated using the clonogenic assay. Apoptosis, cell cycle analysis, reactive oxygen species, mitochondrial membrane potential and caspase-3/- 7 were assessed by flow cytometry. Glutathione level was detected by colorimetric assay. PT-induced alteration in pro-apoptotic/ anti-apoptotic proteins and other signaling pathways were investigated using western blotting. P38 downregulation was performed using siRNA.
Results: In the present study, we explored the molecular mechanism of PT-mediated inhibition of cell proliferation in colorectal cancer cells. PT significantly inhibited the colony formation in human colorectal cancer cell lines (HT-29, SW480 and SW620) by inducing apoptosis and necrosis. This platinum complex was shown to significantly increase the reactive oxygen species (ROS) generation, depletion of glutathione and reduced mitochondrial membrane potential in colorectal cancer cells. Exposure to PT resulted in the downregulation of anti-apoptotic proteins (Bcl2, BclxL, XIAP) and alteration in Cyclins expression. Furthermore, PT increased cytochrome c release into cytosol and enhanced PARP cleavage leading to activation of intrinsic apoptotic pathway. Moreover, pre-treatment with ROS scavenger N-acetylcysteine (NAC) attenuated apoptosis suggesting that PT-induced apoptosis was driven by oxidative stress. Additionally, we show that PT-induced apoptosis was mediated by activating p38 MAPK and inhibiting AKT pathways. This was demonstrated by using chemical inhibitor and siRNA against p38 kinase which blocked the cytochrome c release and apoptosis in colorectal cancer cells.
Conclusion: Collectively, our data demonstrates that the platinum complex (PT) exerts its anti-proliferative effect on CRC by ROS-mediated apoptosis and activating p38 MAPK pathway. Thus, our findings reveal a novel mechanism of action for PT on colorectal cancer cells and may have therapeutic implication.
Keywords: Apoptosis; Colorectal Cancer; MAPK; Platinum; Redox balance.
Conflict of interest statement
All the authors declare no competing interests.
Figures





References
-
- Keum N, Giovannucci E. Global burden of colorectal cancer: emerging trends, risk factors and prevention strategies. Nat Rev Gastroenterol Hepatol. 2019; 10.1038/s41575-019-0189-8. - PubMed
-
- International Agency for Research on Cancer. Globocan 2018: Cancer Fact Sheets — Colorectal Cancer. IARC. 2018. http://gco.iarc.fr/today/data/factsheets/cancers/10_8_9-Colorectum-fact-....
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

