Effect of a quality improvement package for intrapartum and immediate newborn care on fresh stillbirth and neonatal mortality among preterm and low-birthweight babies in Kenya and Uganda: a cluster-randomised facility-based trial
- PMID: 32710862
- PMCID: PMC7388203
- DOI: 10.1016/S2214-109X(20)30232-1
Effect of a quality improvement package for intrapartum and immediate newborn care on fresh stillbirth and neonatal mortality among preterm and low-birthweight babies in Kenya and Uganda: a cluster-randomised facility-based trial
Abstract
Background: Although gains in newborn survival have been achieved in many low-income and middle-income countries, reductions in stillbirth and neonatal mortality have been slow. Prematurity complications are a major driver of stillbirth and neonatal mortality. We aimed to assess the effect of a quality improvement package for intrapartum and immediate newborn care on stillbirth and preterm neonatal survival in Kenya and Uganda, where evidence-based practices are often underutilised.
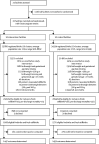
Methods: This unblinded cluster-randomised controlled trial was done in western Kenya and eastern Uganda at facilities that provide 24-h maternity care with at least 200 births per year. The study assessed outcomes of low-birthweight and preterm babies. Eligible facilities were pair-matched and randomly assigned (1:1) into either the intervention group or the control group. All facilities received maternity register data strengthening and a modified WHO Safe Childbirth Checklist; facilities in the intervention group additionally received provider mentoring using PRONTO simulation and team training as well as quality improvement collaboratives. Liveborn or fresh stillborn babies who weighed between 1000 g and 2500 g, or less than 3000 g with a recorded gestational age of less than 37 weeks, were included in the analysis. We abstracted data from maternity registers for maternal and birth outcomes. Follow-up was done by phone or in person to identify the status of the infant at 28 days. The primary outcome was fresh stillbirth and 28-day neonatal mortality. This trial is registered with ClinicalTrials.gov, NCT03112018.
Findings: Between Oct 1, 2016, and April 30, 2019, 20 facilities were randomly assigned to either the intervention group (n=10) or the control group (n=10). Among 5343 eligible babies in these facilities, we assessed outcomes of 2938 newborn and fresh stillborn babies (1447 in the intervention and 1491 in the control group). 347 (23%) of 1491 infants in the control group were stillborn or died in the neonatal period compared with 221 (15%) of 1447 infants in the intervention group at 28 days (odds ratio 0·66, 95% CI 0·54-0·81). No harm or adverse effects were found.
Interpretation: Fresh stillbirth and neonatal mortality among low-birthweight and preterm babies can be decreased using a package of interventions that reinforces evidence-based practices and invests in health system strengthening.
Funding: Bill & Melinda Gates Foundation.
Copyright © 2020 The Author(s). Published by Elsevier Ltd. This is an Open Access article under the CC BY 4.0 license. Published by Elsevier Ltd.. All rights reserved.
Figures

Comment in
-
Implementing change for facility-based peripartum care in low-income and middle-income countries.Lancet Glob Health. 2020 Aug;8(8):e980-e981. doi: 10.1016/S2214-109X(20)30306-5. Lancet Glob Health. 2020. PMID: 32710869 Free PMC article. No abstract available.
-
Respectful language in intrapartum and newborn care.Lancet Glob Health. 2021 Jan;9(1):e17. doi: 10.1016/S2214-109X(20)30462-9. Lancet Glob Health. 2021. PMID: 33338452 No abstract available.
-
Respectful language in intrapartum and newborn care - Authors' reply.Lancet Glob Health. 2021 Jan;9(1):e18. doi: 10.1016/S2214-109X(20)30465-4. Lancet Glob Health. 2021. PMID: 33338453 No abstract available.
References
-
- Lawn JE, Blencowe H, Waiswa P. Stillbirths: rates, risk factors, and acceleration towards 2030. Lancet. 2016;387:587–603. - PubMed
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

