Second-line treatment in advanced gastric cancer: Data from the Spanish AGAMENON registry
- PMID: 32735623
- PMCID: PMC7394396
- DOI: 10.1371/journal.pone.0235848
Second-line treatment in advanced gastric cancer: Data from the Spanish AGAMENON registry
Abstract
Background: Second-line treatments boost overall survival in advanced gastric cancer (AGC). However, there is a paucity of information as to patterns of use and the results achieved in actual clinical practice.
Materials and methods: The study population comprised patients with AGC in the AGAMENON registry who had received second-line. The objective was to describe the pattern of second-line therapies administered, progression-free survival following second-line (PFS-2), and post-progression survival since first-line (PPS).
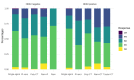
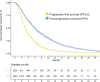
Results: 2311 cases with 2066 progression events since first-line (89.3%) were recorded; 245 (10.6%) patients died during first-line treatment and 1326/2066 (64.1%) received a second-line. Median PFS-2 and PPS were 3.1 (95% CI, 2.9-3.3) and 5.8 months (5.5-6.3), respectively. The most widely used strategies were monoCT (56.9%), polyCT (15.0%), ramucirumab+CT (12.6%), platinum-reintroduction (8.3%), trastuzumab+CT (6.1%), and ramucirumab (1.1%). PFS-2/PPS medians gradually increased in monoCT, 2.6/5.1 months; polyCT 3.4/6.3 months; ramucirumab+CT, 4.1/6.5 months; platinum-reintroduction, 4.2/6.7 months, and for the HER2+ subgroup in particular, trastuzumab+CT, 5.2/11.7 months. Correlation between PFS since first-line and OS was moderate in the series as a whole (Kendall's τ = 0.613), lower in those subjects who received second-line (Kendall's τ = 0.539), especially with ramucirumab+CT (Kendall's τ = 0.413).
Conclusion: This analysis reveals the diversity in second-line treatment for AGC, highlighting the effectiveness of paclitaxel-ramucirumab and, for a selected subgroup of patients, platinum reintroduction; both strategies endorsed by recent clinical guidelines.
Conflict of interest statement
Dr. Javier Gallego declares advisory role for Amgen, Bayer, BMS, Ipsen, Lilly, Merck, Roche, Servier, and travel grants from Novartis, Amgen. No other authors have competing interests to declare. This does not alter our adherence to PLOS ONE policies on sharing data and materials.
Figures


References
-
- Bang YJ, Van Cutsem E, Feyereislova A, Chung HC, Shen L, Sawaki A, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): A phase 3, open-label, randomised controlled trial. Lancet. 2010;376:687–97. 10.1016/S0140-6736(10)61121-X - DOI - PubMed
-
- Yamada Y, Boku N, Mizusawa J, Iwasa S, Kadowaki S, Nakayama N, et al. Docetaxel Plus Cisplatin and S-1 versus Cisplatin and S-1 in Patients with Advanced Gastric Cancer (JCOG1013): An Open-Label, Randomised, Phase 3 Trial. Lancet Gastroenterol Hepatol. 2019;4:501 10.1016/S2468-1253(19)30083-4 - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

