Expanding Role of Dopaminergic Inhibition in Hypercapnic Responses of Cultured Rat Carotid Body Cells: Involvement of Type II Glial Cells
- PMID: 32751703
- PMCID: PMC7432366
- DOI: 10.3390/ijms21155434
Expanding Role of Dopaminergic Inhibition in Hypercapnic Responses of Cultured Rat Carotid Body Cells: Involvement of Type II Glial Cells
Abstract
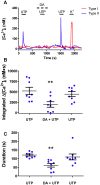
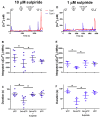
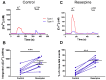
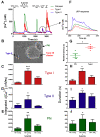
Dopamine (DA) is a well-studied neurochemical in the mammalian carotid body (CB), a chemosensory organ involved in O2 and CO2/H+ homeostasis. DA released from receptor (type I) cells during chemostimulation is predominantly inhibitory, acting via pre- and post-synaptic dopamine D2 receptors (D2R) on type I cells and afferent (petrosal) terminals respectively. By contrast, co-released ATP is excitatory at postsynaptic P2X2/3R, though paracrine P2Y2R activation of neighboring glial-like type II cells may boost further ATP release. Here, we tested the hypothesis that DA may also inhibit type II cell function. When applied alone, DA (10 μM) had negligible effects on basal [Ca2+]i in isolated rat type II cells. However, DA strongly inhibited [Ca2+]i elevations (Δ[Ca2+]i) evoked by the P2Y2R agonist UTP (100 μM), an effect opposed by the D2/3R antagonist, sulpiride (1-10 μM). As expected, acute hypercapnia (10% CO2; pH 7.4), or high K+ (30 mM) caused Δ[Ca2+]i in type I cells. However, these stimuli sometimes triggered a secondary, delayed Δ[Ca2+]i in nearby type II cells, attributable to crosstalk involving ATP-P2Y2R interactions. Interestingly sulpiride, or DA store-depletion using reserpine, potentiated both the frequency and magnitude of the secondary Δ[Ca2+]i in type II cells. In functional CB-petrosal neuron cocultures, sulpiride potentiated hypercapnia-induced Δ[Ca2+]i in type I cells, type II cells, and petrosal neurons. Moreover, stimulation of type II cells with UTP could directly evoke Δ[Ca2+]i in nearby petrosal neurons. Thus, dopaminergic inhibition of purinergic signalling in type II cells may help control the integrated sensory output of the CB during hypercapnia.
Keywords: carotid body; dopamine; petrosal neurons; purinergic signalling; sulpiride; type II cells.
Conflict of interest statement
The authors declare no competing interests.
Figures

References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

