Sonic hedgehog signalling as a potential endobronchial biomarker in COPD
- PMID: 32767976
- PMCID: PMC7412648
- DOI: 10.1186/s12931-020-01478-x
Sonic hedgehog signalling as a potential endobronchial biomarker in COPD
Abstract
Background: The hedgehog (HH) pathway has been associated with chronic obstructive pulmonary disease (COPD) in genome-wide association studies and recent studies suggest that HH signalling could be altered in COPD. We therefore used minimally invasive endobronchial procedures to assess activation of the HH pathway including the main transcription factor, Gli2, and the ligand, Sonic HH (Shh).
Methods: Thirty non-COPD patients and 28 COPD patients were included. Bronchial brushings, bronchoalveolar lavage fluid (BALF) and bronchial biopsies were obtained from fiberoptic bronchoscopy. Characterization of cell populations and subcellular localization were evaluated by immunostaining. ELISA and RNAseq analysis were performed to identify Shh proteins in BAL and transcripts on lung tissues from non-COPD and COPD patients with validation in an external and independent cohort.
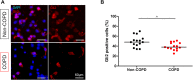
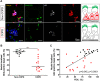
Results: Compared to non-COPD patients, COPD patients exhibited a larger proportion of basal cells in bronchial brushings (26 ± 11% vs 13 ± 6%; p < 0.0001). Airway basal cells of COPD subjects presented less intense nuclear staining for Gli2 in bronchial brushings and biopsies (p < 0.05). Bronchial BALF from COPD patients contained lower Shh concentrations than non-COPD BALF (12.5 vs 40.9 pg/mL; p = 0.002); SHH transcripts were also reduced in COPD lungs in the validation cohort (p = 0.0001).
Conclusion: This study demonstrates the feasibility of assessing HH pathway activation in respiratory samples collected by bronchoscopy and identifies impaired bronchial epithelial HH signalling in COPD.
Keywords: Airway epithelial cells; Bronchoscopy; Chronic obstructive pulmonary disease; Hedgehog signalling pathway.
Conflict of interest statement
Dr. Deslée reports personal fees from Nuvaira, personal fees from BTG/PneumRx, personal fees from Chiesi, personal fees from Boehringer, personal fees from Astra Zeneca, outside the submitted work. Dr. Dury reports personal fees from Novartis, personal fees from Boehringer-Ingelheim, personal fees from Chiesi, personal fees from Roche, outside the submitted work. Dr. Dormoy reports personal fees from Chiesi outside the submitted work.
Figures




References
-
- Diaz-Guzman E, Mannino DM. Epidemiology and prevalence of chronic obstructive pulmonary disease. Clin Chest Med. 2014;35:7–16. - PubMed
-
- Jones RL, Noble PB, Elliot JG, James AL. Airway remodelling in COPD: It’s not asthma! Respirol Carlton Vic. 2016;21:1347–1356. - PubMed
-
- Siafakas N, Corlateanu A, Fouka E. Phenotyping before starting treatment in COPD? COPD. 2017;14:367–374. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

