Labeling a TCO-functionalized single domain antibody fragment with 18F via inverse electron demand Diels Alder cycloaddition using a fluoronicotinyl moiety-bearing tetrazine derivative
- PMID: 32773089
- PMCID: PMC7458057
- DOI: 10.1016/j.bmc.2020.115634
Labeling a TCO-functionalized single domain antibody fragment with 18F via inverse electron demand Diels Alder cycloaddition using a fluoronicotinyl moiety-bearing tetrazine derivative
Abstract
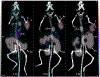
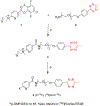
Single domain antibody fragments (sdAbs) exhibit a rapid tumor uptake and fast blood clearance amenable for labeling with 18F (t½ = 110 min) but suffer from high kidney accumulation. Previously, we developed a method for 18F-labeling of sdAbs via trans-cyclooctene (TCO)-tetrazine (Tz) inverse electron demand Diel's Alder cycloaddition reaction (IEDDAR) that incorporated a renal brush border enzyme (RBBE)-cleavable linker. Although >15 fold reduction in kidney activity levels was achieved, tumor uptake was compromised. Here we investigate whether replacing the [18F]AlF-NOTA moiety with [18F]fluoronicotinyl would rectify this problem. Anti-HER2 sdAb 5F7 was first derivatized with a TCO-containing agent that included the RBBE-cleavable linker GlyLys (GK) and a PEG chain, and then subjected to IEDDAR with 6-[18F]fluoronicotinyl-PEG4-methyltetrazine to provide [18F]FN-PEG4-Tz-TCO-GK-PEG4-5F7 ([18F]FN-GK-5F7). For comparisons, a control lacking GK linker and 5F7 labeled using residualizing N-succinimidyl 3-guanidinomethyl-5-[125I]iodobenzoate (iso-[125I]SGMIB) also were synthesized. Radiochemical purity, affinity (KD) and immunoreactive fraction of [18F]FN-GK-5F7 were 99%, 5.4 ± 0.7 nM and 72.5 ± 4.3%, respectively. Tumor uptake of [18F]FN-GK-5F7 in athymic mice bearing subcutaneous SKOV3 xenografts (3.7 ± 1.2% ID/g and 3.4 ± 1.0% ID/g at 1 h and 3 h, respectively) was 2- to 3-fold lower than for co-injected iso-[125I]SGMIB-5F7 (6.9 ± 1.9 %ID/g and 8.7 ± 3.0 %ID/g). However, due to its 6-fold lower kidney activity levels, tumor-to-kidney ratios for [18F]FN-GK-5F7 were 3-4 times higher than those for co-injected iso-[125I]SGMIB-5F7 as well as those observed for the 18F conjugate lacking the RBBE-cleavable linker. Micro-PET/CT imaging of [18F]FN-GK-5F7 in mice with SKOV-3 subcutaneous xenografts clearly delineated tumor as early as 1 h with minimal activity in the kidneys; however, there was considerable activity in gallbladder and intestines. Although the tumor uptake of [18F]FN-GK-5F7 was unexpectedly disappointing, incorporating an alternative RBBE-cleavable linker into this labeling strategy may ameliorate this problem.
Keywords: Fluorine-18; HER2; Inverse electron-demand Diels-Alder reaction; PET imaging; Renal brush border enzyme-cleavable linker; Single domain antibody fragment (VHH, Nanobody).
Copyright © 2020 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest Michael R. Zalutsky is the founder of Cereius, Inc., a company that develops radiolabeling methodologies, including those described herein, for biomolecules including sdAbs. Ganesan Vaidyanathan is a consultant and shareholder of Cereius, Inc.
Figures
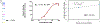
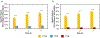
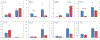
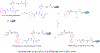
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

