Hematopoietic recovery in patients receiving chimeric antigen receptor T-cell therapy for hematologic malignancies
- PMID: 32780846
- PMCID: PMC7422135
- DOI: 10.1182/bloodadvances.2020002509
Hematopoietic recovery in patients receiving chimeric antigen receptor T-cell therapy for hematologic malignancies
Abstract
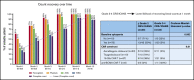
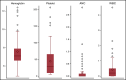
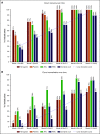
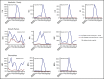
Factors contributing to hematopoietic recovery following chimeric antigen receptor (CAR) T-cell therapy have not been well studied. In an analysis of 83 patients with hematologic malignancies treated with CAR T-cell therapy, we describe patterns of hematopoietic recovery and evaluate potentially associated factors. We included patients who received axicabtagene ciloleucel (n = 30) or tisagenlecleucel (n = 10) for B-cell lymphoma, CD19-28z CAR T therapy for B-cell acute lymphoblastic leukemia (NCT01044069; n = 37), or B-cell maturation antigen targeting CAR T cells for multiple myeloma (NCT03070327; n = 6). Patients treated with CAR T cells who had not progressed, died, or received additional chemotherapy had "recovered" (per definition in Materials and methods section) hemoglobin, platelet, neutrophil, and white blood cell counts at rates of 61%, 51%, 33%, and 28% at month 1 postinfusion and 93%, 90%, 80%, and 59% at month 3 postinfusion, respectively. Univariate analysis showed that increasing grade of immune effector cell-associated neurological syndrome (ICANS), baseline cytopenias, CAR construct, and higher peak C-reactive protein or ferritin levels were statistically significantly associated with a lower likelihood of complete count recovery at 1 month; a similar trend was seen for cytokine release syndrome (CRS). After adjustment for baseline cytopenia and CAR construct, grade ≥3 CRS or ICANS remained significantly associated with the absence of complete count recovery at 1 month. Higher levels of vascular endothelial growth factor and macrophage-derived chemokines, although not statistically significant, were seen patients without complete count recovery at 1 month. This remains to be studied further in larger prospective studies.
© 2020 by The American Society of Hematology.
Conflict of interest statement
Conflict-of-interest disclosures: T.J. has received consulting fees from Takeda Oncology and has served on an advisory board for CareDx. G.L.S. has received research funding from Amgen and Janssen Pharmaceuticals. M. Scordo has served as a paid consultant for McKinsey & Company, Angiocrine Bioscience, Inc., and Omeros Corporation; has received research funds from Angiocrine Bioscience, Inc.; and has served on an ad hoc advisory board for Kite, a Gilead Company. C.S.S. has served as a paid consultant on advisory boards for Juno Therapeutics, Sanofi-Genzyme, Spectrum Pharmaceuticals, Novartis, Genmab, Precision Biosciences, Kite, a Gilead Company, Celgene, Gamida Cell, and GSK and has received research funds for clinical trials from Juno Therapeutics, Celgene, Precision Biosciences, and Sanofi-Genzyme. B.D.S. has acted as a consultant for Kite/Gilead, Juno/Celgene, and Novartis. M.L.P. has acted as a consultant for Noble Insights and served on advisory board for Phamacyclics. An immediate family member has served as consultant for Merck & Co., Inc.; has served on advisory boards for STRAXIMM Therapeutics, Kite Pharmaceuticals, and Seres Therapeutics; has served as a member of the Speakers Bureau for Hemedicus; has equity ownership in Seres Therapeutics and Evelo Biosciences; and holds patents and royalties for Memorial Sloan Kettering Cancer Center (intellectual property for Juno and Seres Therapeutics). C.W.B. has acted as a consultant for and served on advisory boards for Juno Therapeutics. S.G. has acted as a consultant for and received research funding from Amgen, Actinium, Celgene, Johnson & Johnson, and Takeda; has acted as a consultant for Jazz Pharmaceuticals, Novartis, Kite, and Spectrum Pharmaceuticals; and has received research funding from Miltenyi Biotec. R.B. has acted as a consultant for and has patents, royalties, and research funding from Juno Therapeutics and has acted as a consultant for Celgene. E.S. has acted as a consultant for and has patents, royalties, and research funding from Celgene and has acted as a consultant for Fate Therapeutics and Precision Biosciences. J.H.P. has acted as a consultant for Allogene Therapeutics, Amgen, AstraZeneca, Autolus Therapeutics, GSK, Incyte, Kite Pharma, Novartis, and Takeda. M.-A.P. has served on advisory boards for MolMed, NexImmune, Medigene, and Servier; has received honoraria from and served on advisory boards for AbbVie, Bellicum Pharmaceuticals, Bristol-Meyers Squibb, Nektar Therapeutics, Novartis, Omeros, and Takeda; has acted as a consultant for and received honoraria from Merck; and has received research funding from Kite/Gilead, Incyte, and Miltenyi Biotec. S.M. has received research funding from Juno Therapeutics, Celgene, Janssen Pharmaceuticals, Allogene Therapeutics, and Takeda Oncology and has received honoraria for Continuing Medical Education activity from Physician Education Resource and PleXus Education. The remaining authors declare no competing financial interests.
Figures

References
-
- Schuster SJ, Bishop MR, Tam CS, et al. ; JULIET Investigators . Tisagenlecleucel in adult relapsed or refractory diffuse large B-cell lymphoma. N Engl J Med. 2019;380(1):45-56. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

