Multifaceted aspects of charge transfer
- PMID: 32785306
- PMCID: PMC7544685
- DOI: 10.1039/d0cp01556c
Multifaceted aspects of charge transfer
Abstract
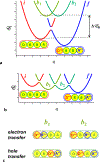
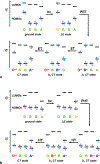
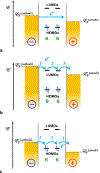
Charge transfer and charge transport are by far among the most important processes for sustaining life on Earth and for making our modern ways of living possible. Involving multiple electron-transfer steps, photosynthesis and cellular respiration have been principally responsible for managing the energy flow in the biosphere of our planet since the Great Oxygen Event. It is impossible to imagine living organisms without charge transport mediated by ion channels, or electron and proton transfer mediated by redox enzymes. Concurrently, transfer and transport of electrons and holes drive the functionalities of electronic and photonic devices that are intricate for our lives. While fueling advances in engineering, charge-transfer science has established itself as an important independent field, originating from physical chemistry and chemical physics, focusing on paradigms from biology, and gaining momentum from solar-energy research. Here, we review the fundamental concepts of charge transfer, and outline its core role in a broad range of unrelated fields, such as medicine, environmental science, catalysis, electronics and photonics. The ubiquitous nature of dipoles, for example, sets demands on deepening the understanding of how localized electric fields affect charge transfer. Charge-transfer electrets, thus, prove important for advancing the field and for interfacing fundamental science with engineering. Synergy between the vastly different aspects of charge-transfer science sets the stage for the broad global impacts that the advances in this field have.
Conflict of interest statement
Conflicts of interest
There are no conflicts to declare
Figures

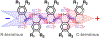


Similar articles
-
On the Search of a Silver Bullet for the Preparation of Bioinspired Molecular Electrets with Propensity to Transfer Holes at High Potentials.Biomolecules. 2021 Mar 15;11(3):429. doi: 10.3390/biom11030429. Biomolecules. 2021. PMID: 33804209 Free PMC article.
-
Resolving Chemical Dynamics in Biological Energy Conversion: Long-Range Proton-Coupled Electron Transfer in Respiratory Complex I.Acc Chem Res. 2021 Dec 21;54(24):4462-4473. doi: 10.1021/acs.accounts.1c00524. Epub 2021 Dec 13. Acc Chem Res. 2021. PMID: 34894649 Free PMC article.
-
Charge transport in nanoscale junctions.J Phys Condens Matter. 2008 Sep 3;20(37):370301. doi: 10.1088/0953-8984/20/37/370301. Epub 2008 Aug 6. J Phys Condens Matter. 2008. PMID: 21694407
-
The Minderoo-Monaco Commission on Plastics and Human Health.Ann Glob Health. 2023 Mar 21;89(1):23. doi: 10.5334/aogh.4056. eCollection 2023. Ann Glob Health. 2023. PMID: 36969097 Free PMC article. Review.
-
Charge migration and charge transfer in molecular systems.Struct Dyn. 2017 Dec 27;4(6):061508. doi: 10.1063/1.4996505. eCollection 2017 Nov. Struct Dyn. 2017. PMID: 29333473 Free PMC article. Review.
Cited by
-
Twisting, Hydrogen Bonding, Exciplex Formation, and Charge Transfer. What Makes a Bright Fluorophore Not So Bright?J Org Chem. 2025 Jul 18;90(28):9714-9732. doi: 10.1021/acs.joc.5c00492. Epub 2025 Jun 4. J Org Chem. 2025. PMID: 40464098 Free PMC article.
-
On the Search of a Silver Bullet for the Preparation of Bioinspired Molecular Electrets with Propensity to Transfer Holes at High Potentials.Biomolecules. 2021 Mar 15;11(3):429. doi: 10.3390/biom11030429. Biomolecules. 2021. PMID: 33804209 Free PMC article.
-
Potent strategy towards strongly emissive nitroaromatics through a weakly electron-deficient core.Chem Sci. 2021 Sep 29;12(42):14039-14049. doi: 10.1039/d1sc03670j. eCollection 2021 Nov 3. Chem Sci. 2021. PMID: 34760187 Free PMC article.
-
Role of intramolecular hydrogen bonds in promoting electron flow through amino acid and oligopeptide conjugates.Proc Natl Acad Sci U S A. 2021 Mar 16;118(11):e2026462118. doi: 10.1073/pnas.2026462118. Proc Natl Acad Sci U S A. 2021. PMID: 33707214 Free PMC article.
-
Photophysics and Electrochemistry of Biomimetic Pyranoflavyliums: What Can Bioinspiration from Red Wines Offer?Photochem. 2022 Mar;2(1):9-31. doi: 10.3390/photochem2010003. Epub 2022 Jan 6. Photochem. 2022. PMID: 35075451 Free PMC article.
References
-
- Vullev VI, J. Phys. Chem. Lett, 2011, 2, 503–508.
-
- Derr JB, Tamayo J, Espinoza EM, Clark JA and Vullev VI, Can. J. Chem, 2018, 96, 843–858.
-
- Pooley DT, QJM, 2010, 103, 545–554. - PubMed
-
- Maeda K, Henbest KB, Cintolesi F, Kuprov I, Rodgers CT, Liddell PA, Gust D, Timmel CR and Hore PJ, Nature, 2008, 453, 387–U338. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

