Engineered Nanoparticle Applications for Recombinant Influenza Vaccines
- PMID: 32787280
- PMCID: PMC8011859
- DOI: 10.1021/acs.molpharmaceut.0c00383
Engineered Nanoparticle Applications for Recombinant Influenza Vaccines
Abstract
Influenza viruses cause seasonal epidemics and represent a pandemic risk. With current vaccine methods struggling to protect populations against emerging strains, there is a demand for a next-generation flu vaccine capable of providing broad protection. Recombinant biotechnology, combined with nanomedicine techniques, could address this demand by increasing immunogenicity and directing immune responses toward conserved antigenic targets on the virus. Various nanoparticle candidates have been tested for use in vaccines, including virus-like particles, protein and carbohydrate nanoconstructs, antigen-carrying lipid particles, and synthetic and inorganic particles modified for antigen presentation. These methods have yielded some promising results, including protection in animal models against antigenically distinct influenza strains, production of antibodies with broad reactivity, and activation of potent T cell responses. Based on the evidence of current research, it is feasible that the next generation of influenza vaccines will combine recombinant antigens with nanoparticle carriers.
Keywords: antigens; influenza; nanoparticles; particles; vaccine.
Figures


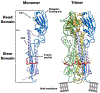




References
-
- Center for Disease Control and Prevention 2019–2020 U.S. Flu Season: Preliminary Burden Estimates https://www.cdc.gov/flu/about/burden/preliminary-in-season-estimates.htm (retrieved June 2020),
-
- Hoffmann E; Krauss S; Perez D; Webby R; Webster RG Eight-plasmid system for rapid generation of influenza virus vaccines. Vaccine 2002, 20, (25), 3165–3170. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

