Systematic Interrogation of Angiogenesis in the Ischemic Mouse Hind Limb: Vulnerabilities and Quality Assurance
- PMID: 32787524
- PMCID: PMC7505144
- DOI: 10.1161/ATVBAHA.120.315028
Systematic Interrogation of Angiogenesis in the Ischemic Mouse Hind Limb: Vulnerabilities and Quality Assurance
Abstract
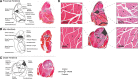
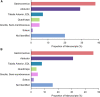
Objective: There has been little success in translating preclinical studies of mouse hind limb ischemia into benefit for patients with peripheral artery disease. Using systematic strategies, we sought to define the injury and angiogenesis landscapes in mice subjected to hind limb ischemia and ascertain whether published studies to date have used an analysis strategy concordant with these data. Approach and Results: Maps of ischemic injury were generated from 22 different hind limb muscles and 33 muscle territories in 12-week-old C57BL/6 mice, based on loss or centralization of myofiber nuclei. Angiogenesis was similarly mapped based on CD (cluster of differentiation) 31-positive capillary content. Only 10 of 33 muscle territories displayed consistent muscle injury, with the distal anterior hind limb muscles most reliably injured. Angiogenesis was patchy and exclusively associated with zones of regenerated muscle (central nuclei). Angiogenesis was not observed in normal appearing muscle, necrotic muscle, or injury border zones. Systematic review of mouse hind limb angiogenesis studies identified 5147 unique publications, of which 509 met eligibility criteria for analysis. Only 7% of these analyzed manuscripts evaluated angiogenesis in distal anterior hind limb muscles and only 15% consistently examined for angiogenesis in zones of muscle regeneration.
Conclusions: In 12-week C57BL/6 mice, angiogenesis postfemoral artery excision proceeds exclusively in zones of muscle regeneration. Only a minority of studies to date have analyzed angiogenesis in regions of demonstrably regenerating muscle or in high-likelihood territories. Quality assurance standards, informed by the atlas and mapping data herein, could augment data reliability and potentially help translate mouse hind limb ischemia studies to patient care.
Keywords: animals; femoral artery; ischemia; lower extremity; muscle, skeletal.
Conflict of interest statement
None.
Figures






References
-
- GBD 2013 Mortality and Causes of Death Collaborators. Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990-2013: a systematic analysis for the global burden of disease study 2013. Lancet. 2015;385:117–171. doi: 10.1016/S0140-6736(14)61682-2 - PMC - PubMed
-
- Fowkes FG, Rudan D, Rudan I, Aboyans V, Denenberg JO, McDermott MM, Norman PE, Sampson UK, Williams LJ, Mensah GA, et al. Comparison of global estimates of prevalence and risk factors for peripheral artery disease in 2000 and 2010: a systematic review and analysis. Lancet. 2013;382:1329–1340. doi: 10.1016/S0140-6736(13)61249-0 - PubMed
-
- Farber A. Chronic limb-threatening ischemia. N Engl J Med. 2018;379:171–180. doi: 10.1056/NEJMcp1709326 - PubMed
-
- Farber A, Eberhardt RT. The current state of critical limb ischemia: a systematic review. JAMA Surg. 2016;151:1070–1077. doi: 10.1001/jamasurg.2016.2018 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

