Subtype and grade-dependent spatial heterogeneity of T-cell infiltration in pediatric glioma
- PMID: 32788236
- PMCID: PMC7422651
- DOI: 10.1136/jitc-2020-001066
Subtype and grade-dependent spatial heterogeneity of T-cell infiltration in pediatric glioma
Abstract
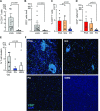
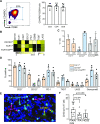
Brain tumors are the leading cause of cancer-related mortality in children and have distinct genomic and molecular features compared with adult glioma. However, the properties of immune cells in these tumors has been vastly understudied compared with their adult counterparts. We combined multiplex immunofluorescence immunohistochemistry coupled with machine learning and single-cell mass cytometry to evaluate T-cells infiltrating pediatric glial tumors. We show that low-grade tumors are characterized by greater T-cell density compared with high-grade glioma (HGG). However, even among low-grade tumors, T-cell infiltration can be highly variable and subtype-dependent, with greater T-cell density in pleomorphic xanthoastrocytoma and ganglioglioma. CD3+ T-cell infiltration correlates inversely with the expression of SOX2, an embryonal stem cell marker commonly expressed by glial tumors. T-cells within both HGG and low-grade glioma (LGG) exhibit phenotypic heterogeneity and tissue-resident memory T-cells consist of distinct subsets of CD103+ and TCF1+ cells that exhibit distinct spatial localization patterns. TCF1+ T-cells are located closer to the vessels while CD103+ resident T-cells reside within the tumor further away from the vasculature. Recurrent tumors are characterized by a decline in CD103+ tumor-infiltrating T-cells. BRAFV600E mutation is immunogenic in children with LGG and may serve as a target for immune therapy. These data provide several novel insights into the subtype-dependent and grade-dependent changes in immune architecture in pediatric gliomas and suggest that harnessing tumor-resident T-cells may be essential to improve immune control in glioma.
Keywords: brain neoplasms; pediatrics; t-lymphocytes; tumor microenvironment.
© Author(s) (or their employer(s)) 2020. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: None declared.
Figures


References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
