Neural and Homeostatic Regulation of REM Sleep
- PMID: 32793050
- PMCID: PMC7385183
- DOI: 10.3389/fpsyg.2020.01662
Neural and Homeostatic Regulation of REM Sleep
Abstract
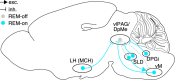
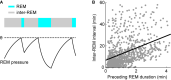
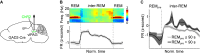
Rapid eye movement (REM) sleep is a distinct, homeostatically controlled brain state characterized by an activated electroencephalogram (EEG) in combination with paralysis of skeletal muscles and is associated with vivid dreaming. Understanding how REM sleep is controlled requires identification of the neural circuits underlying its initiation and maintenance, and delineation of the homeostatic processes regulating its expression on multiple timescales. Soon after its discovery in humans in 1953, the pons was demonstrated to be necessary and sufficient for the generation of REM sleep. But, especially within the last decade, researchers have identified further neural populations in the hypothalamus, midbrain, and medulla that regulate REM sleep by either promoting or suppressing this brain state. The discovery of these populations was greatly facilitated by the availability of novel technologies for the dissection of neural circuits. Recent quantitative models integrate findings about the activity and connectivity of key neurons and knowledge about homeostatic mechanisms to explain the dynamics underlying the recurrence of REM sleep. For the future, combining quantitative with experimental approaches to directly test model predictions and to refine existing models will greatly advance our understanding of the neural and homeostatic processes governing the regulation of REM sleep.
Keywords: REM sleep; REM sleep homeostasis; brain state; neural circuits and behavior; sleep.
Copyright © 2020 Park and Weber.
Figures


References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

