Metformin versus the combined oral contraceptive pill for hirsutism, acne, and menstrual pattern in polycystic ovary syndrome
- PMID: 32794179
- PMCID: PMC7437400
- DOI: 10.1002/14651858.CD005552.pub3
Metformin versus the combined oral contraceptive pill for hirsutism, acne, and menstrual pattern in polycystic ovary syndrome
Abstract
Background: Metformin has been proposed as possibly a safer and more effective long-term treatment than the oral contraceptive pill (OCP) in women with polycystic ovary syndrome (PCOS). It is important to directly compare the efficacy and safety of metformin versus OCP in the long-term treatment of women with PCOS. This is an update of a Cochrane Review comparing insulin sensitising agents with the OCP and only includes studies on metformin.
Objectives: To assess the effectiveness and safety of metformin versus the OCP (alone or in combination) in improving clinical, hormonal, and metabolic features of PCOS.
Search methods: In August 2019 we searched the Cochrane Gynaecology and Fertility Group Trials Register, Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, Embase and CINAHL, the trial registers, handsearched references of the identified articles, and contacted experts in the field to identify additional studies.
Selection criteria: We included randomised controlled trials (RCTs) of the use of metformin versus the OCP (alone or in combination) for women with PCOS.
Data collection and analysis: We used standard methods recommended by Cochrane. The primary review outcomes were the clinical parameters of hirsutism and adverse events, both severe (requiring stopping of medication), and minor. In the presence of substantial heterogeneity (I2 statistic > 50), which could be explained by pre-specified subgroup analyses on the basis of BMI, we reported the subgroups separately.
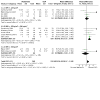
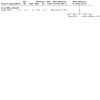
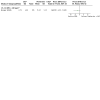
Main results: This is a substantive update. We identified 38 additional studies. We included 44 RCTs (2253 women), which comprised 39 RCTs on adult women (2047 women) and five RCTs on adolescent women (206 women). Evidence quality ranged from very low to low. The main limitations were risk of bias, imprecision and inconsistency. Metformin versus the OCP In adult women, we are uncertain of the effect of metformin compared to the OCP on hirsutism in subgroup body mass index (BMI) < 25 kg/m2 (mean difference (MD) 0.38, 95% confidence interval (CI) -0.44 to 1.19, 3 RCTs, n = 134, I2 = 50%, very low-quality evidence) and subgroup BMI > 30 kg/m2 (MD -0.38, 95% CI -1.93 to 1.17; 2 RCTs, n = 85, I2 = 34%, low-quality evidence). Metformin may be less effective in improving hirsutism compared to the OCP in the subgroup BMI 25 kg/m2 to 30 kg/m2 (MD 1.92, 95% CI 1.21 to 2.64, 5 RCTs, n = 254, I2 = 0%, low-quality evidence). Metformin may increase severe gastro-intestinal adverse events rate compared to the OCP (Peto odds ratio (OR) 6.42, 95% CI 2.98 to 13.84, 11 RCTs, n = 602, I2 = 0%, low-quality evidence). Metformin may decrease the incidence of severe other adverse events compared to the OCP (Peto OR 0.20, 95% CI 0.09 to 0.44, 8 RCTs, n = 363, I2 = 0%, low-quality evidence). There were no trials reporting on minor adverse events. In adolescents, we are uncertain whether there is a difference between Metformin and the OCP, on hirsutism and adverse events. Metformin versus metformin combined with the OCP In adult women, metformin may be less effective in improving hirsutism compared to Metformin combined with the OCP (MD 1.36, 95% CI 0.62 to 2.11, 3 RCTs, n = 135, I2= 9%, low-quality evidence). We are uncertain if there was a difference between metformin and metformin combined with the OCP for severe gastro-intestinal adverse events (OR 0.74, 95% CI 0.21 to 2.53, 3 RCTs, n = 171, I2 = 0%, low-quality evidence), or for severe other adverse events (OR 0.56, 95% CI 0.11 to 2.82, 2 RCTs, n = 109, I2 = 44%, low-quality evidence). There were no trials reporting on minor adverse events. In adolescents, there were no trials for this comparison. The OCP versus metformin combined with the OCP In adult women, the OCP may be less effective in improving hirsutism compared to metformin combined with the OCP (MD 0.54, 95% CI 0.20 to 0.89, 6 RCTs, n = 389, I2= 1%, low-quality evidence). The OCP may decrease the incidence of severe gastro-intestinal adverse events compared to metformin combined with the OCP (OR 0.20, 95% CI 0.06 to 0.72, 5 RCTs, n = 228, I2 = 0%, low-quality evidence). We are uncertain if there is a difference between the OCP and metformin combined with the OCP for severe other adverse events (OR 1.61, 95% CI 0.49 to 5.37, 4 RCTs, n = 159, I2 = 12%, low-quality evidence). The OCP may decrease the incidence of minor (gastro-intestinal) adverse events compared to metformin combined with the OCP (OR 0.06, 95% CI 0.01 to 0.44, 2 RCTs, n = 98, I2 = 0%, low-quality evidence). In adolescents, we are uncertain whether there is a difference between the OCP, compared to metformin combined with the OCP, on hirsutism or adverse events.
Authors' conclusions: In adult women with PCOS, metformin may be less effective in improving hirsutism compared to the OCP in the subgroup BMI 25 kg/m2 to 30 kg/m2 but we are uncertain if there was a difference between metformin and the OCP in subgroups BMI < 25 kg/m2 and BMI > 30kg/m2. Compared to the OCP, metformin may increase the incidence of severe gastro-intestinal adverse events and decrease the incidence of severe other adverse events with no trials reporting on minor adverse events. Either metformin alone or the OCP alone may be less effective in improving hirsutism compared to metformin combined with the OCP. We are uncertain whether there is a difference between the OCP alone and metformin alone compared to metformin combined with the OCP for severe or minor adverse events except for the OCP versus metformin combined with the OCP where the OCP may decrease the incidence of severe and minor gastro-intestinal adverse events. In adolescent women with PCOS, we are uncertain whether there is a difference between any of the comparisons for hirsutism and adverse events due to either no evidence or very low-quality evidence. Further large well-designed RCTs that stratify for BMI are needed to evaluate metformin versus the OCP and combinations in women with PCOS, in particular adolescent women.
Copyright © 2020 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd.
Conflict of interest statement
M Costello has declared shares in Virtus Health and past sponsorship from Merck Serono for scientific conference presentations.
E Fraison has no conflicts of interest to declare.
L Moran has received a National Heart Foundation Future Leader Fellowship that has funded her work on this review.
S Bilal has no conflicts of interest to declare.
C Ee declares that as a medical research institute, NICM Health Research Institute receives research grants and donations from foundations, universities, government agencies and industry. Sponsors and donors provide untied and tied funding for work to advance the vision and mission of the institute. She confirms that she personally received no industry funding relating to this review.
E Kostova has no conflicts of interest to declare.
C Venetis reports an early career Fellowship from National Health and Medical Research Council of Australia which supported him for this work, personal fees and non‐financial support from Merck Sharpe & Dohme, Merck, Beisins, research grants and non‐financial support from Ferring, outside the submitted work.
Figures
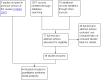



















































































Update of
-
Insulin-sensitising drugs versus the combined oral contraceptive pill for hirsutism, acne and risk of diabetes, cardiovascular disease, and endometrial cancer in polycystic ovary syndrome.Cochrane Database Syst Rev. 2007 Jan 24;(1):CD005552. doi: 10.1002/14651858.CD005552.pub2. Cochrane Database Syst Rev. 2007. Update in: Cochrane Database Syst Rev. 2020 Aug 13;8:CD005552. doi: 10.1002/14651858.CD005552.pub3. PMID: 17253562 Updated.
References
References to studies included in this review
Aghamohammadzadeh 2010 {published data only}
-
- Aghamohammadzadeh N, Aliasgarzadeh A, Baglar L, Abdollahifard S, Bahrami A, Najafipour F, et al. Comparison of metformin and cyproterone-estrodiol compound effect on HS C-reactive protein and serum androgen levels in patients with polycystic ovary syndrome. Pakistan Journal of Medical Science 2010;26(2):347-51.
Allen 2005 {published data only}
-
- Allen HF, Mazzoni C, Heptulla RA, Murray MA, Miller N, Koenigs L, et al. Randomized controlled trial evaluating response to Metformin versus standard therapy in the treatment of adolescents with polycystic ovary syndrome. Journal of Pediatric Endocrinology and Metabolism 2005;18:761-8. - PubMed
Al‐Zubeidi 2015 {published data only}
-
- Al-Zubeidi H, Klein KO. Randomized clinical trial evaluating Metformin versus oral contraceptive pills in the treatment of adolescents with polycystic ovarian syndrome. Journal of Pediatric Endocrinology and Metabolism 2015;28:853-8. - PubMed
Bhattacharya 2016 {published data only}
-
- Bhattacharya S M, Biswas B, Basu A. Comparative study of the therapeutic effects of drospirenone (DRSP, 3mg) with ethinyl estradiol (EE, 20 mcg) pill with or without metformin 500 mg in polycystic ovary syndrome (PCOS). In: Fertility and Sterility. 2016, poster P-401:e257.
Bodur 2018 {published data only}
-
- Bodur S, Dundar O, Kanat-Pektas M, Kinci MF, Tutuncu L. The effects of different therapeutic modalities on cardiovascular risk factors in women with polycystic ovary syndrome: a randomized controlled study. Taiwanese Journal of Obstetrics and Gynecology 2018;57:411-6. - PubMed
Cetinkalp 2009 {published data only}
-
- Cetinkalp S, Karadeniz M, Erdogan M, Ozgen G, Saygili F, Yilmaz C. The effects of rosiglitazone, metformin, and estradiol-cyproterone acetate on lean patients with polycystic ovary syndrome. Endocrinologist 2009;19:94-7.
Christakou 2014 {published data only}
-
- Christakou C, Kollias A, Piperi C, Katsikis I, Panidis D, Diamanti-Kandarakis E. The benefit-to-risk ratio of common treatments in PCOS: effect of oral contraceptives versus metformin on atherogenic markers. Hormones 2014;13:488-97. - PubMed
Cibula 2005 {published data only}
-
- Cibula D, Fanta M, Vrbikova J, Stanicka S, Dvorakova K, Hill M, et al. The effect of combination therapy with metformin and combined oral contraceptives (COC) versus COC alone on insulin sensitivity, hyperandrogenaemia, SHBG and lipids in PCOS patients. Human Reproduction 2005;20(1):180-4. - PubMed
Dardzinska 2014 {published data only}
-
- Dardzinska JA, Rachon D, Kuligowska-Jakuboswka M, Aleksandrowicz-Wrona E, Ploszynski A, Wyrzykowski B, et al. Effects of metformin or an oral contraceptive containing cyproterone acetate on serum C-reactive protein, Interleukine-6 and Soluble Vascular Cell Adhesion Molecule-1 concentrations in women with polycystic ovary syndrome. Experimental and Clinical Endocrinology & Diabetes 2014;122:118-25. - PubMed
El Maghraby 2015 {published data only}
-
- El Maghraby HA, Nafee T, Guiziry D, Elnashar A. Randomized controlled trial of the effects of metformin versus combined oral contraceptives in adolescent PCOS women through a 24 months follow up period. Middle East Fertility Society Journal 2015;20:131-7.
Elter 2002 {published data only}
-
- Elter K, Imir G, Durmusoglu F. Clinical, endocrine and metabolic effects of metformin added to ethinyl-estradiol-cyproterone acetate in non-obese women with polycystic ovarian syndrome: a randomized controlled study. Human Reproduction 2002;17(7):1729-37. - PubMed
Essah 2011 {published data only}
Feng 2016 {published data only}
-
- Feng W, Jia Y-Y, Zhang D-Y, Shi H-R. Management of polycystic ovarian syndrome with Diane-35 or Diane-35 plus metformin. Gynecological Endocrinology 2016;32(2):147-50. - PubMed
Glintborg 2014a {published data only}
-
- Glintborg D, Altinok ML, Mumm H, Hermann AP, Ravn P, Andersen M. Body composition is improved during 12 month's treatment with Metformin alone or combined with oral contraceptives compared with treatment with oral contraceptives in polycystic ovary syndrome. Endocrine Research 2014;99:2584-91. - PubMed
Harborne 2003 {published and unpublished data}
-
- Harborne L, Fleming R, Lyall H, Sattar N, Norman J. Metformin or antiandrogen in the treatment of hirsutism in polycystic ovary syndrome. Journal of Clinical Endocrinology & Metabolism 2003;88(9):4116-23. - PubMed
Hoeger 2008a {published data only}
-
- Hoeger K, Davidson K, Kochman L, Cherry T, Kopin L, Guzick S. The impact of metformin, oral contraceptives, and lifestyle modification on polycystic ovary syndrome in obese adolescent women in two randomized, placebo-controlled clinical trials. Journal of Clinical Endocrinology and Metabolism 2008;93:4299-306. - PMC - PubMed
Hoeger 2008b {published data only}
-
- Hoeger K, Davidson K Kochman L, Cherry T, Kopin L, Guzick S. The impact of metformin, oral contraceptives, and lifestyle modification on polycystic ovary syndrome in obese adolescent women in two randomized, placebo-controlled clinical trials. Journal of Clinical Endocrinology and Metabolism 2008;93:4299-306. - PMC - PubMed
Jin 2006 {published data only}
-
- Jin LN, Yu Q, Ma L K. The effect of combination therapy with Metformin and combined Cyproterone Acetate/ Ethinyl Estradiol in PCOS patients. Chinese Journal of Postgraduate Medicine 2006;29:10-5.
Kaya 2015 {published data only}
-
- Kaya MG, Yildirim S, Calapkorur B, Akpek M, Unluhizarci K, Kelestimur F. Metformin improves endothelial function and carotid intima media thickness in patients with PCOS. Gynecological Endocrinology 2015;31:401-5. - PubMed
Kebapcilar 2009a {published data only}
-
- Kebapcilar L, Taner CE, Kebapcilar AG, Alacacioglu A, Sari I. Comparison of four different treatment regimens on coagulation parameters, hormonal and metabolic changes in women with polycystic ovary syndrome. Archives of Gynecology and Obstetrics 2009;281:35-42. - PubMed
Kebapcilar 2009b {published data only}
-
- Bilgir O, Kebapcilar L, Taner C, Bilgir F, Kebapcilar A, Bozkaya G, et al. The effect of ethinylestradiol (EE)/cyproterone acetate (CA) and EE/CA plus metformin treatment on adhesion molecules in cases with polycystic ovary syndrome (PCOS). International Medicine 2009;48:1193-9. - PubMed
-
- Kebapcilar L, Yuksel A, Bozkaya G, Taner CE, Kebapcilar AG, Bilgir O, et al. Effects of an EE/CA compared with EE/CA-metformin on serum ADMA levels in women with polycystic ovary syndrome. Central European Journal of Medicine 2009;4:423-7.
Kilic 2011 {published data only}
-
- Kilic S, Yilmaz N, Zulfikaroglu E, Erdogan G, Aydin M, Batioglu S. Inflammatory-metabolic parameters in obese and nonobese normoandrogenemic polycystic ovary syndrome during metformin and oral contraceptive treatment. Gynecological Endocrinology 2011;27(9):622-9. - PubMed
Kuek 2011 {published data only}
-
- Kuek S, Wang W-J, Gui S-Q. Efficacy of chinese patent medicine Tian Gui Capsule in patients with polycystic ovary syndrome: a randomized controlled trial. Journal of Chinese Integrative Medicine 2011;9:965-72. - PubMed
Kumar 2018 {published data only}
Liu 2006 {published data only}
-
- Liu JX, Dang JM, Wu YZ, Zheng N, Wu XK. The effect of combination therapy with Metformin and combined Cyproterone Acetate/Ethynil Estradiol on PCOS patients. Chinese Journal of Postgraduate Medicine 2006;19:530-3.
Luque‐Ramirez 2007a {published data only}
-
- Luque-Ramirez M, Alvarez-Blasco F, Botella-Carretero JI, Martinez-Bermejo E, Lasuncion MA, Escobar-Morreale HF. Comparison of ethinyl-estradiol plus cyproterone acetate versus metformin effects on classic metabolic cardiovascular risk factors in women with polycystic ovary syndrome. Journal of Clinical Endocrinology and Metabolism 04/2007;92:2453-61. - PubMed
Luque‐Ramirez 2007b {published data only}
-
- Luque-Ramirez M, Alvarez-Blasco F, Botella-Carretero J I, Sanchon R, San Millan JL, Escobar-Morreale HF. Increased body iron stores of obese women with polycystic ovary syndrome are a consequence of insulin resistance and hyperinsulinism and are not a result of reduced menstrual losses. Diabetes Care 09/2007;30:2309-13. - PubMed
Luque‐Ramirez 2008a {published data only}
-
- Luque-Ramirez M, Alvarez-Blasco F, Uriol Rivera MG, Escobar-Morreale HF. Serum uric acid concentration as non-classic cardiovascular risk factor in women with polycystic ovary syndrome: effect of treatment with ethinyl-estradiol plus cyproterone acetate versus metformin. Human Reproduction 2008;23:1594-601. - PubMed
Luque‐Ramirez 2009b {published data only}
-
- Luque-Ramirez M, Mendieta-Azcona C, Alvarez-Blasco F, Escobar-Morreale HF. Effects of Metformin versus Ethinyl-estradiol plus cyproterone acetate on ambulatory blood pressure monitoring and carotid intima media thickness in women with the polycystic ovary syndrome. Fertility and Sterility 2008;91:2527-36. - PubMed
Lv 2005 {published data only}
-
- Lv L, Liu Y, Sun Y, Tan K. Effects of Metformin combined with cyproterone acetate on clinical features, endocrine and metabolism of non-obese women with polycystic ovarian syndrome. Journal of Huazhong University of Science and Technology 2005;25:194-7. - PubMed
Meyer 2007 {published data only}
-
- Meyer C, McGrath BP, Teede HJ. Effect of medical therapy on insulin resistance and the cardiovascular system in polycystic ovary syndrome. Diabetes Care 2007;30(3):471-8. - PubMed
Mhao 2015 {published data only}
-
- Mhao NS, Al-Hilli SA, Hadi NR, Jamil DA, Al-Aubaidy HA. A comparative study to illustrate the benefits of using ethinyl estradiol-cyproterone acetate over metformin in patients with polycystic ovarian syndrome. Diabetes and Metabolic Syndrome: Clinical Research and Reviews 2015;10S:S95-S98. - PubMed
Moran 2010 {published data only}
-
- Moran LJ, Meyer C, Hutchison SK, Zoungas S, Teede HJ. Novel inflammatory markers in overweight women with or without polycystic ovary syndrome and following pharmacological intervention. Journal of Endocrinological Investigation 2010;33:258-65. - PubMed
Morin‐Papunen 2000 {published data only}
-
- Morin-Papunen LC, Vauhkonen I, Koivunen RM, Ruokonen A, Martikainen HK, Tapanainen JS. Endocrine and metabolic effects of metformin versus ethinyl-estradiol-cyproterone acetate in obese women with polycystic ovary syndrome: a randomized study. Journal of Clinical Endocrinology & Metabolism 2000;85(9):3161-8. - PubMed
Morin‐Papunen 2003 {published data only}
-
- Morin-Papunen L, Vauhkonen I, Koivunen R, Ruokonen A, Martikainen H, Tapanainen JS. Metformin versus ethinyl estradiol-cyproterone acetate in the treatment of nonobese women with polycystic ovary syndrome: a randomized study. The Journal of Clinical Endocrinology & Metabolism 2003;88(1):148-56. - PubMed
Moro 2013 {published data only}
-
- Moro F, Morciano A, Tropea A, Sagnella F, Palla C, Scarinci E, et al. Effects of drospirenone-ethinylestradiol and/or metformin on CD4+ CD28 null T lymphocytes frequency in women with hyperinsulinemia having polycystic ovary syndrome: a randomized clinical trial. Reproductive Science 2013;20(12):1508-17. - PubMed
Ozgurtas 2008 {published data only}
-
- Ozgurtas T, Oktenli C, Dede M, Tapan S, Kenar L, Sanisoglu SY, et al. Metformin and oral contraceptive treatment reduced circulating asymmetric dimethylarginine (ADMA) levels in patients with polycystic ovary syndrome (PCOS). Atherosclerosis 2008;200:336-44. - PubMed
Rautio 2005 {published data only}
-
- Rautio K, Tapanainen JS, Ruokonen A, Morin-Papunen LC. Effects of metformin and ethinyl-estradiol-cyproterone acetate on lipid levels in obese and non-obese women with polycystic ovary syndrome. European Journal of Endocrinology 2005;152:269-75. - PubMed
Ruan 2018 {published data only}
-
- Ruan X, Song J, Gu M, Wang L, Wang H, Mueck AO. Effect of Diane-35, alone or in combination with orlistat or metformin in Chinese polycystic ovary syndrome patients. Archives of Gynecology and Obstetrics 2018;297:1557-63. - PubMed
Sahu 2018 {published data only}
-
- Sahu A, Tripathy P, Mohanty J, Nagy A. Doppler analysis of ovarian stromal blood flow changes after treatment with metformin versus ethinyl estradiol-cyproterone acetate in women with polycystic ovarian syndrome: a randomized controlled trial. Journal of Gynecology Obstetrics and Human Reproduction 2018;48(5):335-9. - PubMed
Song 2017 {published data only}
-
- Song J, Ruan X, Gu M, Wang L, Wang H, Mueck AO. Effect of orlistat or metformin in overweight and obese polycystic ovary syndrome patients with insulin resistance. Gynecological Endocrinology 2018;34:413-7. - PubMed
Teng 2007 {published data only}
-
- Teng YQ, Zheng JH, Han XY. Evaluation of clinical efficacy of Metformin in treating polycystic ovary syndrome. Journal of Harbin Medical University 2007;41:405-6.
Wei 2012 {published data only}
-
- Wei W, Zhao H, Wang A, Sui M, Liang K, Deng H, et al. A clinical study on the short-term effect of berberine in comparison to metformin on the metabolic characteristics of women with polycystic ovary syndrome. European Journal of Endocrinology 2012;166:99-105. - PubMed
Wu 2008 {published data only}
-
- Wu J, Zhu Y, Jiang Y, Cao Y. Effects of metformin and ethinyl estradiol-cyproterone acetate on clinical, endocrine and metabolic factors in women with polycystic ovary syndrome. Gynecological Endocrinology 2008;24:392-8. - PubMed
References to studies excluded from this review
Alpanes 2017 {published data only}
-
- Alpanes M, Alvarez-Blasco F, Fernandez-Duran E, Luque-Ramirez M, Escobar-Morreale HF. Combined oral contraceptives plus spironolactone compared with Metformin in women with polycystic ovary syndrome: a one-year randomized clinical trial. European Journal of Endocrinology 2017;177:399-408. - PubMed
Altinok 2018 {published data only}
-
- Altinok ML, Ravn P, Andersen M, Glintoborg D. Effect of 12-months treatment with metformin and/or oral contraceptives on health-related quality of life in polycystic ovary syndrome. Gynecological Endocrinology 2018;34(10):859-63. - PubMed
Bachani 2016 {published data only}
-
- Bachani S, Ganju Malla V. Identifying risk factors and screening tests for extent of metabolic risk in urban adolescent girls in a developing nation. International journal of child and adolescent health 2016;9(3):347-53.
Bhattacharya 2012 {published data only}
-
- Battacharya SM, Jha A. Comparative study of the therapeutic effects of oral contraceptive pills containing desogestrel, cyproterone acetate, and drospirenone in patients with polycystic ovary syndrome. Fertility and sterility 2012;98(4):1053-9. - PubMed
Bredella 2013 {published data only}
Burchall 2015 {published data only}
-
- Burchall GF, Piva TJ, Linden MD, Gibson-Helm ME, Ranasinha S, Teede HJ. Comprehensive assessment of the hemostatic system in polycystic ovarian syndrome. Seminars in Thrombosis and Hemostasis 2016;42:55-62. - PubMed
Cakiroglu 2013 {published data only}
-
- Cakiroglu Y, Vural B, Isgoren S. The effects of drospirenone-ethinyl estradiol and drospirenone-ethinyl estradiol + metformin on ovarian ultrasonographic markers, body fat mass index, leptin, and ghrelin. Archive of Gynecology and Obstetrics 2013;288:213-20. - PubMed
Diaz 2016 {published data only}
-
- Diaz M, Gallego-Escuredo JM, Zegher F. Effect of ethinylestradiol-cyproterone acetate vs. pioglitazone-flutamide-metformin on plasma FGF21 levels in adolescent girls with androgen excess. Diabetes and Metabolism 2016;42:196-9. - PubMed
Glintborg 2014b {published data only}
-
- Glintborg D, Mumm H, Altinok ML, Richelsen B, Bruun JM, Andersen M. Adiponectin, interleukin-6, monocyte chemoattractant protein-1, and regional fat mass during 12-months randomized treatment with metformin and/or contraceptives in polycystic ovary syndrome. Journal of Endocrinal Investigation 2014;37:757-64. - PubMed
Glintborg 2015 {published data only}
-
- Glintborg D, Sidelmann JJ, Altinok ML, Mumm H, Andersen M. Increased thrombin generation in women with polycystic ovary syndrome. A pilot study on the effect of metformin and oral contraceptives. Metabolism 2015;64:1272-8. - PubMed
Glintborg 2017 {published data only}
Hadziomerovic‐Pekic 2010 {published data only}
-
- Hadziomerovic-Pekic D, Wildt L, Weiss JM, Moeller K, Mattle V, Seeber BE. Metformin, naltrexone, or the combination of prednisolone and antiandrogenic oral contraceptives as first-line therapy in hyperinsulinemic women with polycystic ovary syndrome. Fertility and Sterility 2010;94(6):2385-8. - PubMed
Harris‐Glocker 2009 {published data only}
-
- Harris-Glocker M, Davidson K, Kochman L, Guzick D, Hoeger K. Improvement in quality of life questionnaire measures (PCOSQ) in obese adolescent females with PCOS treated with Lifestyle changes and oral contraceptives, with or without Metformin. Fertility and Sterility 2009;93(3):1016-9. - PMC - PubMed
Hu 2010 {published data only}
-
- Hu Z, Wang Y, Qiao J, Li M, Chi H, Chen X. The role of family history in clinical symptoms and therapeutic outcomes of women with polycystic ovary syndrome. International Journal of Gynecology and Obstetrics 2010;108:35-9. - PubMed
Hutchison 2008 {published data only}
Ibanez 2010 {published data only}
-
- Ibanez L, Lopez-Bermejo A, Diaz M, Enriquez G, Rio L, Zegher F. Low-dose pioglitazone, flutamide, metformin plus an oestro-progestagen for non-obese young women with polycystic ovary syndrome: increasing efficacy and persistent safety over 30 months. Gynecological Endocrinology 2010;26(12):869-73. - PubMed
Ibanez 2017 {published data only}
-
- Ibanez L, Rio L, Diaz M, Sebastiani G, Pozo OJ, Lopez-Bermejo A, et al. Normalizing ovulation rate by preferential reduction of hepato-visceral fat in adolescent girls with polycystic ovary syndrome. Journal of Adolescent Health 2017;61:446-53. - PubMed
Kebapcilar 2010 {published data only}
-
- Kebapcilar L, Kebapcilar AG, Bilgir O, Taner CE, Bozkaya G, Yilidiz Y, et al. Metformin plus oral contraceptive may decrease plasma sCD40 ligand in women with PCOS patients. Gynecological Endocrinology 2010;27(2):91-5. - PubMed
Kim 2010 {published data only}
-
- Kim C-H, Jeon G-H, Kim S-R, Kim S-H, Chae H-D, Kang B-M. Effects of pioglitazone on ovarian stromal blood flow, ovarian stimulation, and in vitro fertilization outcome in patients with polycystic ovary syndrome. Fertility and Sterility 2010;94:236-41. - PubMed
Ladson 2011 {published data only}
Lazaro 2011 {published data only}
-
- Lazaro I, Diaz M, Cabre A, Masana L, Ibanez L. Fatty acid-blinding protein-4 plasma levels are associated to metabolic abnormalities and response to therapy in girls and young women with androgen excess. Gynecological Endocrinology 2011;27(11):935-9. - PubMed
Lemay 2006 {published data only}
-
- Lemay A, Dodin S, Turcot L, Déchêne F, Forest J-C. Rosiglitazone and ethinyl estradiol/cyproterone acetate as single and combined treatment of overweight women with polycystic ovary syndrome and insulin resistance. Human Reproduction 2006;21(1):121-8. - PubMed
Luque‐Ramirez 2008c {published data only}
-
- Luque-Ramirez M, Alvarez-Blasco F, Escobar-Morreale HF. Antiandrogenic contraceptives increase serum adiponectin in obese polycystic ovary syndrome patients. Obesity 2008;17(1):3-9. - PubMed
Luque‐Ramirez 2009 {published data only}
-
- Luque-Ramirez M, Mendieta-Azcona C, Rey Sanchez JM, Maties M, Escobar-Morreale HF. Effects of an antiandrogenic oral contraceptive pill compared with metformin on blood coagulation tests and endothelial function in women with the polycystic ovary syndrome: influence of obesity and smoking. European Journal of Endocrinology 2009;160:469-80. - PubMed
Luque‐Ramirez 2010a {published data only}
-
- Luque-Ramirez M, Escobar-Morreale HF. Treatment of polycystic ovary syndrome (PCOS) with metformin ameliorates insulin resistance in parallel with decrease of serum interleukine-6 concentrations. Hormone and Metabolic Research 2010;42:815-20. - PubMed
Luque‐Ramirez 2011 {published data only}
-
- Luque-Ramirez M, Alvarez-Blasco F, Alpanes M, Escobar-Morreale HF. Role of decreased circulating hepcidin concentrations in the iron excess of women with polycystic ovary syndrome. Journal of Clinical Endocrinology and Metabolism 2011;96(3):846-52. - PubMed
Mehrabian 2016 {published data only}
Mitkov 2005 {published data only}
-
- Mitkov M, Pehlivanov B, Terzieva D. Combined use of metformin and ethinyl estradiol-cyproterone acetate in polycystic ovary syndrome. European Journal of Obstetrics and Gynecology and Reproductive Biology 2005;118:209-13. - PubMed
Moghtadaei 2009 {published data only}
-
- Moghtadaei P, Sardari F. Metabolic and endocrine effect of metformin and metformin plus cyclic dydrogestrel in women with polycystic ovarian syndrome. Cell Journal 2009;11:67-8.
Moretti 2016 {published data only}
-
- Moretti CG, Guccione L, di Giacinto P, Cannuccia A, Meleca C, Lanzolla G, et al. Efficacy and safety of myo-inositol supplementation in the treatment of obese hirsute PCOS women: comparative evaluation with OCP+bicalutamide therapy. In: Endocrine Society Journal, poster SUN-153. 2016.
NCT02866786 {published data only}
-
- NCT02866786. The effect of OCP and Metformin on Clinical, hormonal, metabolic and ultrasonographic characteristics in PCOS. clinicaltrials.gov/show/NCT02866786 (first received 15 August 2016).
Orbetzova 2011 {published data only}
-
- Orbetzova MM, Pehlivanov BK, Mitko MM, Atanassova IB, Kamenov ZA, Kolarov GB, et al. Effect of short-term standard therapeutic regimens on neuropeptide Y and adipose tissue hormones in overweight insulin-resistant women with polycystic ovary syndrome. Folia Medica 2011;53(3):15-24. - PubMed
Panidis 2011 {published data only}
-
- Panidis D, Georgopoulos NA, Piouka A, Katsikis I, Saltamavros AD, Decavalas G, et al. The impact of oral contraceptives and metformin on anti-Müllerian hormone serum levels in women with polycystic ovary syndrome and biochemical hyperandrogenemia. Gynecological Endocrinology 2011;27(8):587-92. - PubMed
Pedersen 2018 {published data only}
-
- Pedersen AJ, Stage TB, Glintborg D, Andersen M, Christensen MM. The pharmacogenetics of metformin in women with polycystic ovary syndrome: a randomized trial. Basic and Clinical Pharmacology and Toxicology 2018;122:239-44. - PubMed
Romualdi 2010 {published data only}
-
- Romualdi D, Giuliani M, Cristello F, Fulghesu AM, Selvaggi L, Lanzone A, et sl. Metformin effects on ovarian ultrasound appearance and steroidogenic function in normal-weight normoinsulinemic women with polycystic ovary syndrome; a randomized double-blind placebo-controlled clinical trial. Fertility and Sterility 2010;93(7):2303-10. - PubMed
Suvarna 2016 {published data only}
Teede 2010b {published data only}
-
- Teede HJ, Meyer C, Hutchison SK, Zoungas S, McGrath BP, Moran LJ. Endothelial function and insulin resistance in polycystic ovary syndrome: the effects of medical therapy. Fertility and Sterility 2010;93(1):184-91. - PubMed
Wang 2016 {published data only}
-
- Wang Q-Y, Song Y, Huang W, Xiao L, Wang Q-S, Feng G-M. Comparison of drospirenone with cyproterone acetate containing oral contraceptives, combined with metformin and lifestyle modifications in women with polycystic ovary syndrome and metabolic disorders: a prospective randomized control trial. Chinese Medical Journal 2016;129(8):883-90. - PMC - PubMed
References to studies awaiting assessment
Fruzzeti 2009 {published data only}
-
- Fruzzetti F, Perini D, Lazzarini V, Parrini D, Gambacciani M, Genazzani RA. Comparison of effects of 3 mg drospirenone plus 20 µg ethinyl estradiol alone or combined with metformin or cyproterone acetate on classic metabolic cardiovascular risk factors in nonobese women with polycystic ovary syndrome. Fertility and Sterility 2009;94:1793-8. - PubMed
Spremovic‐Radjenovic 2014 {published data only}
-
- Spremovic-Radjenovic S, Sreckovic S, Gudovic A, Djakonovic M, Bila J, Radonjic-Lazovic G. Comparison of the effects of dianogest/ethinyl estradiol and metformin on metabolic parameters in non-obese women with polycystic ovary syndrome. In: Poster, European Society of Contraception and Reproductive Health, S207. 2014.
Vieira 2010 {published data only}
-
- Vieira CS, Fernandes JB, Soares GM, Martins WP, Silva-de-Sà MF, Ferriani RA. Addition of low dose of metformin to the oral contraceptive has no great advantage in terms of cardiovascular risk markers in women with polycystic ovary syndrome regardless insulin resistance. Fertility and Sterility 2010;94(4 Suppl 1):S63-4. [Oral communication O-217]
References to ongoing studies
NCT02744131 {published data only}
-
- OCP vs metformin for Improvement in clinical symptoms and metabolic markers in Indian PCOS women (OCP). Ongoing study. 04/2016. Contact author for more information.
NCT03229057 {published data only}
-
- Comparing the effects of oral contraceptive pills versus metformin (COMET-PCOS). Ongoing study. Juillet 2017. Contact author for more information.
NCT03905941 {published data only}
-
- Relative desirability of metformin vs. birth control pill in treating PCOS in women of later reproductive age. Ongoing study. March 2020. Contact author for more information.
Additional references
Al Khalifah 2016
-
- Al Khalifah RA, Florez ID, Dennis B, Thsbsnr L, Bassilious E. Metformin or oral contraceptives for adolescents with polycystic ovarian syndrome: a meta-analysis. Peditrics 2016;137(5):e20154089. - PubMed
Buzney 2014
-
- Buzney E, D, Sheu J, Buzney C, Reynolds RV. Polycystic ovary syndrome: a review for dermatologists Part II. Treatment. Journal of the American Academy of Dermatology. 2014;71(5):859.e1-859.e15; quiz 873-4. - PubMed
Conway 1990
-
- Conway GS, Jacobs HS, Holly JM, Wass JA. Effects of luteinizing hormone, insulin, insulin-like growth factor-1 and insulin-like growth factor small binding protein-1 in the polycystic ovary syndrome. Clinical Endocrinology 1990;33:593-603. - PubMed
Costello 2003
-
- Costello MF, Eden JA. A systematic review of the reproductive system effects of Metformin in patients with polycystic ovary syndrome. Fertility and Sterility 2003;79(1):1-13. - PubMed
Deeks 2017
-
- Deeks JJ, Higgins JP, Altman DG. Chapter 9: Analysing data and undertaking meta-analyses. In: Higgins JP, Churchill R, Chandler J, Cumpston MS, editor(s), Cochrane Handbook for Systematic Reviews of Interventions version 5.2.0 (updated June 2017). The Cochrane Collaboration, 2017. Available from www.training.cochrane.org/handbook.
Diamanti‐Kandarakis 2003
-
- Diamanti-Kandarakis E, Baillargeon, JP, Iuorno MJ, Jakubowicz DJ, Nestler JE. A modern medical quandary: polycystic ovary syndrome, insulin resistance, and oral contraceptive pills. Journal of Clinical Endocrinology & Metabolism 2003;88:1927-32. - PubMed
Diamanti‐Kandarakis 2012
Dunaif 1989
-
- Dunaif A, Segal KR, Futterweit W, Dobrjansky A. Profound peripheral insulin resistance, independent of obesity, in polycystic ovary syndrome. Diabetes 1989;38:1165-74. - PubMed
Dunaif 1994
-
- Dunaif A. Molecular mechanisms of insulin resistance in the polycystic ovary syndrome. Seminars in Reproductive Endocrinology 1994;12:15-20.
ESHRE/ASRM 2004
-
- The Rotterdam ESHRE/ASRM-sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Human Reproduction 2004;19:41-7. - PubMed
Fauser 2012
-
- Fauser BC, Tarlatzis BC, Rebar RW, Legro RS, Balen AH, Lobo R, et al. Consensus on women’s health aspects of polycystic ovary syndrome (PCOS): the Amsterdam ESHRE/ ASRM-Sponsored 3rd PCOS Consensus Workshop Group. Fertil Steril 2012;97:28-38.e25. - PubMed
Freitas de Medeiros 2017
Gallo 2013
-
- Gallo MF, Nanda K, Grimes DA, Lopez LM, Schulz KF. 20 µg versus >20 µg estrogen combined oral contraceptives for contraception.. Cochrane Database of Systematic Reviews 2013, Issue 8. Art. No: CD003989. [DOI: 10.1002/14651858.CD003989.pub5] - DOI
GRADEpro GDT [Computer program]
-
- McMaster University (developed by Evidence Prime) GRADEpro GDT. Hamilton (ON): McMaster University (developed by Evidence Prime), accessed 6 August 2019. Available at gradepro.org.
Hasegawa 1999
-
- Hasegawa I, Murakawa H, Suzuki M, Yamamoto Y, Kurabayashi T, Tanaka K. Effect of troglitazone on endocrine and ovulatory performance in women with insulin resistance-related polycystic ovary syndrome. Fertility and Sterility 1999;71:323-7. - PubMed
Higgins 2003
Higgins 2017
-
- Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al. Cochrane Handbook for Systematic Reviews of Interventions version 5.2 (updated 2017). The Cochrane Collaboration, 2011. Available from www.training.cochrane.org/handbook.
Homburg 1996
-
- Homburg R. Polycystic ovary syndrome - from gynaecological curiosity to multisystem endocrinopathy. Human Reproduction 1996;11:29-39. - PubMed
Hundal 2003
-
- Hundal RS, Inzucchi SE. Metformin: new understandings, new uses. Drugs 2003;63(18):1879-94. - PubMed
Katsiki 2010
-
- Katsiki N, Hatzitolios AI. Insulin-sensitizing agents in the treatment of polycystic ovary syndrome: an update. Current Opinion in Obstetrics & Gynecology 2010;22(6):466-76. - PubMed
Luque‐Ramirez 2018
-
- Luque-Ramirez M, Nattero-Chavez L, Ortiz Flores AE, Escobar-Morreale HF. Combined oral contraceptive and/or antiandrogens versus insulin sensitizers for polycystic ovary syndrome: a systematic review and meta-analysis. Human Reproduction Update 2018;24:225-41. - PubMed
Martin 2018
-
- Martin KA, Anderson RR, Chang RJ, Ehrmann DA, Lobo RA, Murad MH, et al. Evaluation and treatment of hirsutism in premenopausal women: an Endocrine Society* Clinical Practice Guideline. Journal of Clinical and Endocrinology Metabolism 2018;103(4):1–25. - PubMed
Ovalle 2002
-
- Ovalle F, Azziz R. Insulin resistance, polycystic ovary syndrome, and type 2 diabetes mellitus. Fertility and Sterility 2002;77:1095-105. - PubMed
Review Manager 2014 [Computer program]
-
- Nordic Cochrane Centre, The Cochrane Collaboration Review Manager 5 (RevMan 5). Version 5.3. Copenhagen: Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Rotterdam ESHRE 2004
-
- The Rotterdam ESHRE/ASRM-sponsored PCOS consensus workshop group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Human Reproduction 2004;19:41-7. - PubMed
Ryan 2016
-
- Ryan R, Cochrane Consumers and Communication Review Group. Cochrane Consumers and Communication Group: meta-analysis. http://cccrg.cochrane.org accessed December 2016.
Teede 2010a
Teede 2018
Teede 2019
-
- Teede H, Tassone EC, Piltonen T, Malhotra J, Mol BW, Pena A, et al. Effect of the combined oral contraceptive pill and/or Metformin in the management of polycystique ovary syndrome: a systematic review with meta-analyses. Clinical Endocrinology 2019;91:479-89. - PubMed
Utiger 1996
-
- Utiger R. Insulin and the polycystic ovary syndrome. New England Journal of Medicine 1996;335:657-8. - PubMed
Wild 2002a
-
- Wild RA. Long-term health consequences of PCOS. Human Reproduction Update 2002;8:231-41. - PubMed
Wild 2002b
-
- Wild RA. Polycystic ovary syndrome: a risk for coronary artery disease? American Journal of Obstetrics & Gynaecology 2002;186:35-43. - PubMed
References to other published versions of this review
Costello 2005
-
- Costello MF, Shrestha B, Eden J, Sjoblom P, Johnson N. Insulin‐sensitising drugs versus the combined oral contraceptive pill for hirsutism, acne, and risk of diabetes, cardiovascular disease, and endometrial cancer in polycystic ovary syndrome. Cochrane Database of Systematic Reviews 2005, Issue 4. Art. No: CD005552. [DOI: 10.1002/14651858.CD005552] - DOI - PubMed
Costello 2007
-
- Costello MF, Shrestha B, Eden J, Johnson N, Moran LJ. Insulin‐sensitising drugs versus the combined oral contraceptive pill for hirsutism, acne and risk of diabetes, cardiovascular disease, and endometrial cancer in polycystic ovary syndrome. Cochrane Database of Systematic Reviews 2007, Issue 1. Art. No: CD005552. [DOI: 10.1002/14651858.CD005552.pub2] - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

