Over-the-Counter Ocular Decongestants in the United States - Mechanisms of Action and Clinical Utility for Management of Ocular Redness
- PMID: 32801982
- PMCID: PMC7399465
- DOI: 10.2147/OPTO.S259398
Over-the-Counter Ocular Decongestants in the United States - Mechanisms of Action and Clinical Utility for Management of Ocular Redness
Abstract
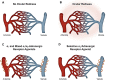
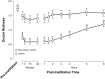
To manage ocular redness effectively, health-care practitioners require an understanding of the pathophysiology, clinical features and differential diagnosis of ocular redness, as well as comprehensive knowledge of medical therapies available and their pharmacologic properties. This review aims to provide a clinically relevant summary of the current literature on the mechanism of action, efficacy, and safety of current over-the-counter (OTC) decongestants available for reduction of ocular redness due to minor irritations. Currently marketed OTC products indicated for such use in the United States include topical solutions of tetrahydrozoline 0.05%, naphazoline 0.012% to 0.03%, and brimonidine 0.025%. All 3 agents are adrenergic receptor agonists but vary in their receptor-binding profiles: tetrahydrozoline is a selective α1 receptor agonist; naphazoline is a mixed α1/α2 receptor agonist; and brimonidine is a selective α2 receptor agonist. These OTC decongestants produce vasoconstriction of conjunctival blood vessels, which results in a rapid reduction in ocular redness. In general, ocular adverse events reported in published studies of these OTC decongestants were minimal, mild, and transient, with no significant adverse systemic effects. However, ocular decongestants with α1-adrenergic receptor agonist activity can be associated with loss of effectiveness with continued use (ie, tachyphylaxis) and rebound redness upon treatment discontinuation. In clinical trials of the selective α2-adrenergic receptor agonist brimonidine 0.025%, tachyphylaxis was not observed, and rebound redness was rarely reported.
Keywords: brimonidine; naphazoline; ocular decongestant; ocular redness; over-the-counter; tetrahydrozoline.
© 2020 Hosten and Snyder.
Conflict of interest statement
LOH and CS are employees of Bausch Health US, LLC. The authors report no other conflicts of interest in this work.
Figures

Similar articles
-
Brimonidine tartrate ophthalmic solution 0.025% for redness relief: an overview of safety and efficacy.Expert Rev Clin Pharmacol. 2022 Aug;15(8):911-919. doi: 10.1080/17512433.2022.2112948. Epub 2022 Aug 18. Expert Rev Clin Pharmacol. 2022. PMID: 35951740 Review.
-
Evaluation of Efficacy and Safety of Brimonidine Tartrate Ophthalmic Solution, 0.025% for Treatment of Ocular Redness.Curr Eye Res. 2018 Jan;43(1):43-51. doi: 10.1080/02713683.2017.1381269. Epub 2017 Nov 9. Curr Eye Res. 2018. PMID: 29120262 Clinical Trial.
-
Brimonidine Ophthalmic Solution 0.025% for Reduction of Ocular Redness: A Randomized Clinical Trial.Optom Vis Sci. 2018 Mar;95(3):264-271. doi: 10.1097/OPX.0000000000001182. Optom Vis Sci. 2018. PMID: 29461408 Free PMC article. Clinical Trial.
-
Low-dose brimonidine for relief of ocular redness: integrated analysis of four clinical trials.Clin Exp Optom. 2019 Mar;102(2):131-139. doi: 10.1111/cxo.12846. Epub 2018 Dec 7. Clin Exp Optom. 2019. PMID: 30525235 Free PMC article. Clinical Trial.
-
The evolving pharmacotherapeutic profile of brimonidine, an alpha 2-adrenergic agonist, after four years of continuous use.Expert Opin Pharmacother. 2000 May;1(4):815-34. doi: 10.1517/14656566.1.4.815. Expert Opin Pharmacother. 2000. PMID: 11249518 Review.
Cited by
-
Safety of Once-Daily Oxymetazoline HCl Ophthalmic Solution, 0.1% in Patients with Acquired Blepharoptosis: Results from Four Randomized, Double-Masked Clinical Trials.Clin Ophthalmol. 2021 Oct 8;15:4035-4048. doi: 10.2147/OPTH.S322326. eCollection 2021. Clin Ophthalmol. 2021. PMID: 34675472 Free PMC article.
-
Ingestion of Fluids of the Ocular Surface Containing Eye Drops of Imidazole Derivatives-Alpha Adrenergic Receptor Agonists as Paragons.Pharmaceuticals (Basel). 2024 Jun 9;17(6):758. doi: 10.3390/ph17060758. Pharmaceuticals (Basel). 2024. PMID: 38931425 Free PMC article. Review.
-
Conceptual Model for Using Imidazoline Derivative Solutions in Pulpal Management.J Clin Med. 2021 Mar 15;10(6):1212. doi: 10.3390/jcm10061212. J Clin Med. 2021. PMID: 33803990 Free PMC article. Review.
-
Ocular complaints and diagnoses in spaceflight.NPJ Microgravity. 2024 Jan 2;10(1):1. doi: 10.1038/s41526-023-00335-7. NPJ Microgravity. 2024. PMID: 38167407 Free PMC article.
-
A Multicenter Randomized Double-Masked Study Comparing Preservative-free Brimonidine Tartrate Ophthalmic Solution 0.025% with LUMIFY® 0.025% for Ocular Redness in Adults.Ophthalmol Ther. 2025 Sep;14(9):2283-2299. doi: 10.1007/s40123-025-01194-z. Epub 2025 Jul 25. Ophthalmol Ther. 2025. PMID: 40711722 Free PMC article.
References
-
- Cronau H, Kankanala RR, Mauger T. Diagnosis and management of red eye in primary care. Am Fam Physician. 2010;81(2):137–144. - PubMed
-
- Adamczyk DT, Jaanus SD. Antiallergy drugs and decongestants In: Bartlett JD, Jaanus SD, editors. Clinical Ocular Pharmacology. 5th ed. St Louis, MO: Butterworth-Heinemann; 2008.
-
- US Food and Drug Administration. 21CFR349.75 Labeling of ophthalmic vasoconstrictor drug products; 2019. Available from: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?f.... Accessed June15, 2020.
Publication types
LinkOut - more resources
Full Text Sources

