Spatiotemporally Controlled Photoresponsive Hydrogels: Design and Predictive Modeling from Processing through Application
- PMID: 32802013
- PMCID: PMC7418561
- DOI: 10.1002/adfm.202000639
Spatiotemporally Controlled Photoresponsive Hydrogels: Design and Predictive Modeling from Processing through Application
Abstract
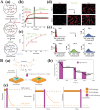
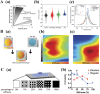
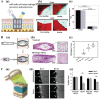
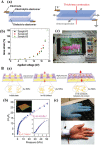
Photoresponsive hydrogels (PRHs) are soft materials whose mechanical and chemical properties can be tuned spatially and temporally with relative ease. Both photo-crosslinkable and photodegradable hydrogels find utility in a range of biomedical applications that require tissue-like properties or programmable responses. Progress in engineering with PRHs is facilitated by the development of theoretical tools that enable optimization of their photochemistry, polymer matrices, nanofillers, and architecture. This review brings together models and design principles that enable key applications of PRHs in tissue engineering, drug delivery, and soft robotics, and highlights ongoing challenges in both modeling and application.
Keywords: hydrogels; mechanical properties; models; photodegradation; photo‐crosslinking.
© 2020 WILEY‐VCH Verlag GmbH & Co. KGaA, Weinheim.
Conflict of interest statement
The authors declare no conflict of interest.
Figures











Similar articles
-
Engineering the Cell Microenvironment Using Novel Photoresponsive Hydrogels.ACS Appl Mater Interfaces. 2018 Apr 18;10(15):12374-12389. doi: 10.1021/acsami.7b17751. Epub 2018 Apr 5. ACS Appl Mater Interfaces. 2018. PMID: 29537822 Review.
-
Recent advances in photo-crosslinkable hydrogels for biomedical applications.Biotechniques. 2019 Jan;66(1):40-53. doi: 10.2144/btn-2018-0083. Biotechniques. 2019. PMID: 30730212 Review.
-
Modeling Controlled Photodegradation in Optically Thick Hydrogels.J Polym Sci A Polym Chem. 2013 May 1;51(9):1899-1911. doi: 10.1002/pola.26574. J Polym Sci A Polym Chem. 2013. PMID: 24496479 Free PMC article.
-
Photoresponsive hydrogels for biomedical applications.Adv Drug Deliv Rev. 2011 Nov;63(14-15):1257-66. doi: 10.1016/j.addr.2011.06.009. Epub 2011 Jul 2. Adv Drug Deliv Rev. 2011. PMID: 21745509 Review.
-
Photoconfigurable, Cell-Remodelable Disulfide Cross-linked Hyaluronic Acid Hydrogels.Biomacromolecules. 2020 Dec 14;21(12):4663-4672. doi: 10.1021/acs.biomac.0c00603. Epub 2020 Sep 3. Biomacromolecules. 2020. PMID: 32830955
Cited by
-
Advances in Injectable In Situ-Forming Hydrogels for Intratumoral Treatment.Pharmaceutics. 2021 Nov 18;13(11):1953. doi: 10.3390/pharmaceutics13111953. Pharmaceutics. 2021. PMID: 34834369 Free PMC article. Review.
-
Synergistic Degradation of Durable Polymer Networks by Light and Acid Enabled by Pyrenylsilicon Crosslinks.Adv Mater. 2025 Jan;37(3):e2412544. doi: 10.1002/adma.202412544. Epub 2024 Dec 4. Adv Mater. 2025. PMID: 39628304 Free PMC article.
-
Anomalous Loss of Stiffness with Increasing Reinforcement in a Photo-Activated Nanocomposite.Macromol Rapid Commun. 2021 Jul;42(14):e2100147. doi: 10.1002/marc.202100147. Epub 2021 May 29. Macromol Rapid Commun. 2021. PMID: 34051002 Free PMC article.
-
Light from Afield: Fast, High-Resolution, and Layer-Free Deep Vat 3D Printing.Chem Rev. 2024 Jul 24;124(14):8787-8822. doi: 10.1021/acs.chemrev.4c00134. Epub 2024 Jul 5. Chem Rev. 2024. PMID: 38967405 Free PMC article. Review.
-
Advanced Multifunctional Hydrogels for Enhanced Wound Healing through Ultra-Fast Selenol-SNAr Chemistry.Adv Sci (Weinh). 2024 Jun;11(21):e2400898. doi: 10.1002/advs.202400898. Epub 2024 Apr 22. Adv Sci (Weinh). 2024. PMID: 38647422 Free PMC article.
References
-
- a) Wallin T. J., Pikul J., Shepherd R. F., Nat. Rev. Mater. 2018, 3, 84;
- b) Truby R. L., Lewis J. A., Nature 2016, 540, 371; - PubMed
- c) Wang R., Sing M. K., Avery R. K., Souza B. S., Kim M., Olsen B. D., Acc. Chem. Res. 2016, 49, 2786; - PubMed
- d) Tomatsu I., Peng K., Kros A., Adv. Drug Delivery Rev. 2011, 63, 1257; - PubMed
- e) Ma Y., Lin M., Huang G., Li Y., Wang S., Bai G., Lu T. J., Xu F., Adv. Mater. 2018, 30, 1705911; - PubMed
- f) Dong Y., Jin G., Hong Y., Zhu H., Lu T. J., Xu F., Bai D., Lin M., ACS Appl. Mater. Interfaces 2018, 10, 12374. - PubMed
-
- Matsuda T., Kawakami R., Namba R., Nakajima T., Gong J. P., Science 2019, 363, 504. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
