Design, Synthesis, Molecular Modeling Study and Biological Evaluation of New N'-Arylidene-pyrido [2,3- d]pyrimidine-5-carbohydrazide Derivatives as Anti-HIV-1 Agents
- PMID: 32802103
- PMCID: PMC7393058
- DOI: 10.22037/ijpr.2019.112198.13597
Design, Synthesis, Molecular Modeling Study and Biological Evaluation of New N'-Arylidene-pyrido [2,3- d]pyrimidine-5-carbohydrazide Derivatives as Anti-HIV-1 Agents
Abstract
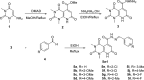
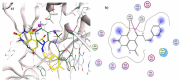
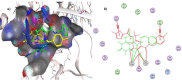
In an attempt to identify potential new agents that are active against HIV-1, a series of novel pyridopyrimidine-5-carbohydrazide derivatives featuring a substituted benzylidene fragment were designed and synthesized based on the general pharmacophore of HIV-1 integrase inhibitors. The cytotoxicity profiles of these compounds showed no significant toxicity to human cells and they exhibited anti-HIV-1 activity with EC50 values ranging from 90 to 155 µM. Compound 5j bearing 4-methylbenzylidene group was found to be the most active compound with EC50 = 90 µM and selectivity index, CC50/EC50 = 6.4. Molecular modeling studies indicated the capacity of compound 5j to interact with two Mg2+ cations and several residues that are important in HIV-1 integrase inhibition. These findings suggested that pyridopyrimidine-5-carbohydrazide scaffold might become a promising template for development of novel anti-HIV-1 agents.
Keywords: Anti-HIV-1; HIV-1 integrase; Molecular modeling; Pyridopyrimidine-5-carbohydrazide; Synthesis.
Figures
Similar articles
-
Synthesis, Biological Evaluation, and Molecular Modeling Studies of New 8-methyl-4-oxo-1,4-dihydroquinoline-3-carbohydrazides as Potential Anti-HIV Agents.Iran J Pharm Res. 2022 May 17;21(1):e123962. doi: 10.5812/ijpr-123962. eCollection 2022 Dec. Iran J Pharm Res. 2022. PMID: 36060911 Free PMC article.
-
Design, Synthesis, Docking Study, and Biological Evaluation of 4-hydroxy-2-oxo-1,2-dihydroquinoline-3-carbohydrazide Derivatives as Anti-HIV-1 and Antibacterial Agents.Iran J Pharm Res. 2022 May 4;21(1):e126562. doi: 10.5812/ijpr-126562. eCollection 2022 Dec. Iran J Pharm Res. 2022. PMID: 36060913 Free PMC article.
-
Design, Synthesis, Molecular Modeling Studies and Biological Evaluation of N'-Arylidene-6-(benzyloxy)-4-oxo-1,4-dihydroquinoline-3-carbohydrazide Derivatives as Novel Anti-HCV Agents.Iran J Pharm Res. 2019 Fall;18(4):1790-1802. doi: 10.22037/ijpr.2019.112186.13586. Iran J Pharm Res. 2019. PMID: 32184846 Free PMC article.
-
Review on the Synthesis and Therapeutic Potential of Pyrido[2,3-d], [3,2-d], [3,4-d] and [4,3-d]pyrimidine Derivatives.Pharmaceuticals (Basel). 2022 Mar 14;15(3):352. doi: 10.3390/ph15030352. Pharmaceuticals (Basel). 2022. PMID: 35337149 Free PMC article. Review.
-
Sketching the historical development of pyrimidones as the inhibitors of the HIV integrase.Eur J Med Chem. 2015 Jun 5;97:649-63. doi: 10.1016/j.ejmech.2014.07.005. Epub 2014 Jul 3. Eur J Med Chem. 2015. PMID: 25084622 Review.
Cited by
-
Synthesis, Biological Evaluation, and Molecular Modeling Studies of New 8-methyl-4-oxo-1,4-dihydroquinoline-3-carbohydrazides as Potential Anti-HIV Agents.Iran J Pharm Res. 2022 May 17;21(1):e123962. doi: 10.5812/ijpr-123962. eCollection 2022 Dec. Iran J Pharm Res. 2022. PMID: 36060911 Free PMC article.
-
Design, Synthesis, Docking Study, and Biological Evaluation of 4-hydroxy-2-oxo-1,2-dihydroquinoline-3-carbohydrazide Derivatives as Anti-HIV-1 and Antibacterial Agents.Iran J Pharm Res. 2022 May 4;21(1):e126562. doi: 10.5812/ijpr-126562. eCollection 2022 Dec. Iran J Pharm Res. 2022. PMID: 36060913 Free PMC article.
-
4-(1-Benzyl-1H-benzo[d]imidazol-2-yl)-4-oxo-2-butenoic Acid Derivatives: Design, Synthesis and Anti-HIV-1 Activity.Iran J Pharm Res. 2021 Winter;20(1):408-417. doi: 10.22037/ijpr.2020.114341.14803. Iran J Pharm Res. 2021. PMID: 34400969 Free PMC article.
-
Design, Synthesis, Docking Study and Biological Evaluation of 4-Hydroxy-2H-benzo[e][1,2]thiazine-3-carboxamide 1,1-dioxide Derivatives as Anti-HIV Agents.Iran J Pharm Res. 2021 Summer;20(3):1-12. doi: 10.22037/ijpr.2020.114153.14695. Iran J Pharm Res. 2021. PMID: 34903964 Free PMC article.
-
Novel 2-(Diphenylmethylidene) Malonic Acid Derivatives as Anti-HIV Agents: Molecular Modeling, Synthesis and Biological Evaluation.Iran J Pharm Res. 2021 Dec 14;21(1):e123827. doi: 10.5812/ijpr.123827. eCollection 2022 Dec. Iran J Pharm Res. 2021. PMID: 35765501 Free PMC article.
References
-
- Organization WH. HIV/AIDS. 2017. [cited; Available from: http://www.who.int/hiv/data/en/
-
- Sun L, Gao P, Dong G, Zhang X, Cheng X, Ding X, Wang X, Daelemans D, De Clercq E, Pannecouque C. 5-Hydroxypyrido [2,3-b] pyrazin-6(5H)-one derivatives as novel dual inhibitors of HIV-1 reverse transcriptase-associated ribonuclease H and integrase. Eur. J. Med. Chem. 2018;155:714–24. - PubMed
-
- Zhan P, Pannecouque C, De Clercq E, Liu X. Anti-HIV drug discovery and development: current innovations and future trends: miniperspective. J. Med. Chem. 2015;59:2849–78. - PubMed
LinkOut - more resources
Full Text Sources
