Bioelectricity for Drug Delivery: The Promise of Cationic Therapeutics
- PMID: 32803148
- PMCID: PMC7313641
- DOI: 10.1089/bioe.2020.0012
Bioelectricity for Drug Delivery: The Promise of Cationic Therapeutics
Abstract
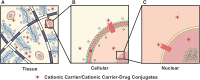
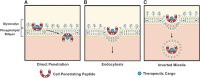
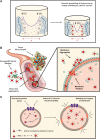
Biological systems overwhelmingly comprise charged entities generating electrical activity that can have significant impact on biological structure and function. This intrinsic bio-electrical activity can also be harnessed for overcoming the tissue matrix and cell membrane barriers, which have been outstanding challenges for targeted drug delivery, by using rationally designed cationic carriers. The weak and reversible long-range electrostatic interactions with fixed negatively charged groups facilitate electro-diffusive transport of cationic therapeutics through full-tissue thickness to effectively reach intra-tissue, cellular, and intracellular target sites. This article presents a perspective on the promise of using rationally designed cationic biomaterials in targeted drug delivery, the underlying charge-based mechanisms, and bio-transport phenomena while addressing outstanding concerns around toxicity and methods to mitigate them. We also discuss electrically charged drugs that are currently being evaluated in clinical trials and identify areas of further development that have the potential to usher in new treatments.
Keywords: cationic drug carriers; cell-penetrating peptides; cytotoxicity; drug delivery; electrostatic interactions; multilevel targeting.
Conflict of interest statement
Author Disclosure Statement No competing financial interests exist.
Figures

Similar articles
-
Cartilage penetrating cationic peptide carriers for applications in drug delivery to avascular negatively charged tissues.Acta Biomater. 2019 Jul 15;93:258-269. doi: 10.1016/j.actbio.2018.12.004. Epub 2018 Dec 6. Acta Biomater. 2019. PMID: 30529083 Free PMC article.
-
Overcoming negatively charged tissue barriers: Drug delivery using cationic peptides and proteins.Nano Today. 2020 Oct;34:100898. doi: 10.1016/j.nantod.2020.100898. Epub 2020 Jun 20. Nano Today. 2020. PMID: 32802145 Free PMC article.
-
Charge-based drug delivery to cartilage: Hydrophobic and not electrostatic interactions are the dominant cause of competitive binding of cationic carriers in synovial fluid.Acta Biomater. 2022 Oct 1;151:278-289. doi: 10.1016/j.actbio.2022.08.010. Epub 2022 Aug 11. Acta Biomater. 2022. PMID: 35963518 Free PMC article.
-
Peptide-Mediated Delivery of Chemical Probes and Therapeutics to Mitochondria.Acc Chem Res. 2016 Sep 20;49(9):1893-902. doi: 10.1021/acs.accounts.6b00277. Epub 2016 Aug 16. Acc Chem Res. 2016. PMID: 27529125 Review.
-
Cartilage-targeting drug delivery: can electrostatic interactions help?Nat Rev Rheumatol. 2017 Mar;13(3):183-193. doi: 10.1038/nrrheum.2016.210. Epub 2017 Feb 9. Nat Rev Rheumatol. 2017. PMID: 28202920 Review.
Cited by
-
Respirators in Healthcare: Material, Design, Regulatory, Environmental, and Economic Considerations for Clinical Efficacy.Glob Chall. 2022 Apr 12;6(10):2200001. doi: 10.1002/gch2.202200001. Online ahead of print. Glob Chall. 2022. PMID: 35601599 Free PMC article. Review.
-
Network Thermodynamical Modeling of Bioelectrical Systems: A Bond Graph Approach.Bioelectricity. 2021 Mar 1;3(1):3-13. doi: 10.1089/bioe.2020.0042. Epub 2021 Mar 16. Bioelectricity. 2021. PMID: 34476374 Free PMC article.
-
Harnessing exosomes for advanced osteoarthritis therapy.Nanoscale. 2024 Oct 24;16(41):19174-19191. doi: 10.1039/d4nr02792b. Nanoscale. 2024. PMID: 39323205 Free PMC article. Review.
-
Charge-Reversed Exosomes for Targeted Gene Delivery to Cartilage for Osteoarthritis Treatment.Small Methods. 2024 Sep;8(9):e2301443. doi: 10.1002/smtd.202301443. Epub 2024 Apr 12. Small Methods. 2024. PMID: 38607953
-
A quality by design approach for the synthesis of palmitoyl-L-carnitine-loaded nanoemulsions as drug delivery systems.Drug Deliv. 2023 Dec;30(1):2179128. doi: 10.1080/10717544.2023.2179128. Drug Deliv. 2023. PMID: 36803136 Free PMC article.
References
-
- Yoon JH, Halper J. Tendon proteoglycans: Biochemistry and function. J Musculoskelet Neuronal Interact 2005;5:22–34 - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
