Off-label intrathecal use of gadobutrol: safety study and comparison of administration protocols
- PMID: 32803338
- PMCID: PMC7803712
- DOI: 10.1007/s00234-020-02519-4
Off-label intrathecal use of gadobutrol: safety study and comparison of administration protocols
Abstract
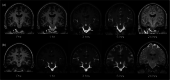
Purpose: Magnetic resonance imaging (MRI) contrast agents have been used off-label for diagnosis of cerebrospinal fluid (CSF) leaks and lately also for assessment of the glymphatic system and meningeal lymphatic drainage. The purpose of this study was to further evaluate the short- and long-term safety profile of intrathecal MRI contrast agents.
Methods: In this prospective study, we compared the safety profile of different administration protocols of intrathecal gadobutrol (GadovistTM; 1.0 mmol/ml). Gadobutrol was administered intrathecal in a dose of 0.5 mmol, with or without iodixanol (VisipaqueTM 270 mg I/ml; 3 ml). In addition, a subgroup was given intrathecal gadobutrol in a dose of 0.25 mmol. Adverse events were assessed at 1 to 3 days, 4 weeks, and after 12 months.
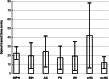
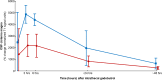
Results: Among the 149 patients, no serious adverse events were seen in patients without history of prior adverse events. The combination of gadobutrol with iodixanol did not increase the occurrence of non-serious adverse events after days 1-3. Intrathecal gadobutrol in a dose of 0.25 mmol caused less severity of nausea, as compared with the dose of 0.5 mmol. The clinical diagnosis was the major determinant for occurrence of non-serious adverse events after intrathecal gadobutrol.
Conclusion: This prospective study showed that intrathecal administration of gadobutrol in a dose of 0.5 mmol is safe. Non-serious adverse events were to a lesser degree affected by the administration protocols, though preliminary data are given that side effects of intrathecal gadobutrol are dose-dependent.
Keywords: Contrast agents; Gadobutrol; Glymphatic function; Iodixanol; Magnetic resonance imaging; Safety.
Conflict of interest statement
Author Geir Ringstad has received a speaker fee from Bayer AG. The other authors declare that they have no conflicts of interest.
Figures
Similar articles
-
Prospective Safety Study of Intrathecal Gadobutrol in Different Doses.AJNR Am J Neuroradiol. 2023 May;44(5):511-516. doi: 10.3174/ajnr.A7841. Epub 2023 Apr 6. AJNR Am J Neuroradiol. 2023. PMID: 37024308 Free PMC article.
-
Intrathecal Use of Gadobutrol for Glymphatic MR Imaging: Prospective Safety Study of 100 Patients.AJNR Am J Neuroradiol. 2019 Aug;40(8):1257-1264. doi: 10.3174/ajnr.A6136. Epub 2019 Jul 18. AJNR Am J Neuroradiol. 2019. PMID: 31320462 Free PMC article.
-
Safety of gadobutrol, a new generation of contrast agents: experience from clinical trials and postmarketing surveillance.Invest Radiol. 2011 Nov;46(11):663-71. doi: 10.1097/RLI.0b013e3182218dc3. Invest Radiol. 2011. PMID: 21623211
-
Safety of Intrathecal Administration of Gadolinium-based Contrast Agents: A Systematic Review and Meta-Analysis.Radiology. 2020 Oct;297(1):75-83. doi: 10.1148/radiol.2020191373. Epub 2020 Jul 28. Radiology. 2020. PMID: 32720867
-
Clinical Safety of Gadobutrol: Review of Over 25 Years of Use Exceeding 100 Million Administrations.Invest Radiol. 2024 Sep 1;59(9):605-613. doi: 10.1097/RLI.0000000000001072. Epub 2024 Mar 1. Invest Radiol. 2024. PMID: 38426761 Review.
Cited by
-
Direction and magnitude of cerebrospinal fluid flow vary substantially across central nervous system diseases.Fluids Barriers CNS. 2021 Apr 1;18(1):16. doi: 10.1186/s12987-021-00251-6. Fluids Barriers CNS. 2021. PMID: 33794929 Free PMC article.
-
Molecular trans-dural efflux to skull bone marrow in humans with CSF disorders.Brain. 2022 May 24;145(4):1464-1472. doi: 10.1093/brain/awab388. Brain. 2022. PMID: 34849609 Free PMC article.
-
Different Glymphatic Kinetics in Spontaneous Intracranial Hypotension.AJNR Am J Neuroradiol. 2024 Oct 3;45(10):1605-1612. doi: 10.3174/ajnr.A8365. AJNR Am J Neuroradiol. 2024. PMID: 39299796
-
Encephalopathy after unintentional intrathecal gadolinium: A letter to the editor.Interv Pain Med. 2022 Jun 17;1(3):100105. doi: 10.1016/j.inpm.2022.100105. eCollection 2022 Sep. Interv Pain Med. 2022. PMID: 39238511 Free PMC article.
-
Clinical application of intrathecal gadobutrol for assessment of cerebrospinal fluid tracer clearance to blood.JCI Insight. 2021 May 10;6(9):e147063. doi: 10.1172/jci.insight.147063. JCI Insight. 2021. PMID: 33822769 Free PMC article.
References
-
- Iliff JJ, Wang M, Liao Y, Plogg BA, Peng W, Gundersen GA, Benveniste H, Vates GE, Deane R, Goldman SA, Nagelhus EA, Nedergaard M. A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid beta. Sci Transl Med. 2012;4(147):147ra111. doi: 10.1126/scitranslmed.3003748. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

