Role of c-Fos in orthodontic tooth movement: an in vivo study using transgenic mice
- PMID: 32803442
- PMCID: PMC7819946
- DOI: 10.1007/s00784-020-03503-1
Role of c-Fos in orthodontic tooth movement: an in vivo study using transgenic mice
Abstract
Objectives: The transcription factor c-Fos controls the differentiation of osteoclasts and is expressed in periodontal ligament cells after mechanical stimulation in vitro. However, it is unclear how c-Fos regulates orthodontic tooth movement (OTM) in vivo. The aim of this study was therefore to analyse OTM in transgenic mice with overexpression of c-Fos.
Materials and methods: We employed c-Fos transgenic mice (c-Fos tg) and wild-type littermates (WT) in a model of OTM induced by Nitinol tension springs that were bonded between the left first maxillary molars and the upper incisors. The unstimulated contralateral side served as an internal control. Mice were analysed by contact radiography, micro-computed tomography, decalcified histology and histochemistry.
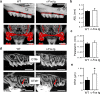
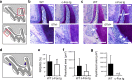
Results: Our analysis of the unstimulated side revealed that alveolar bone and root morphology were similar between c-Fos tg and control mice. However, we observed more osteoclasts in the alveolar bone of c-Fos tg mice as tartrate-resistant acid phosphatase (TRAP)-positive cells were increased by 40%. After 12 days of OTM, c-Fos tg mice exhibited 62% increased tooth movement as compared with WT mice. Despite the faster tooth movement, c-Fos tg and WT mice displayed the same amount of root resorption. Importantly, we did not observe orthodontically induced tissue necrosis (i.e. hyalinization) in c-Fos tg mice, while this was a common finding in WT mice.
Conclusion: Overexpression of c-Fos accelerates tooth movement without causing more root resorption.
Clinical relevance: Accelerated tooth movement must not result in more root resorption as higher tissue turnover may decrease the amount of mechanically induced tissue necrosis.
Keywords: Bone remodelling; Mechanical stimulation; Orthodontic tooth movement; Root resorption; c-Fos.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures



References
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous

