Development of 5D3-DM1: A Novel Anti-Prostate-Specific Membrane Antigen Antibody-Drug Conjugate for PSMA-Positive Prostate Cancer Therapy
- PMID: 32803984
- PMCID: PMC7957835
- DOI: 10.1021/acs.molpharmaceut.0c00457
Development of 5D3-DM1: A Novel Anti-Prostate-Specific Membrane Antigen Antibody-Drug Conjugate for PSMA-Positive Prostate Cancer Therapy
Abstract
Prostate cancer (PC) is a potentially high-risk disease and the most common cancer in American men. It is a leading cause of cancer-related deaths in men in the US, second only to lung and bronchus cancer. Advanced and metastatic PC is initially treated with androgen deprivation therapy (ADT), but nearly all cases eventually progress to castrate-resistant prostate cancer (CRPC). CRPC is incurable in the metastatic stage but can be slowed by some conventional chemotherapeutics and second-generation ADT, such as enzalutamide and abiraterone. Therefore, novel therapeutic strategies are urgently needed. Prostate-specific membrane antigen (PSMA) is overexpressed in almost all aggressive PCs. PSMA is widely used as a target for PC imaging and drug delivery. Anti-PSMA monoclonal antibodies (mAbs) have been developed as bioligands for diagnostic imaging and targeted PC therapy. However, these mAbs are successfully used in PC imaging and only a few have gone beyond phase-I for targeted therapy. The 5D3 mAb is a novel, high-affinity, and fast-internalizing anti-PSMA antibody. Importantly, 5D3 mAb demonstrates a unique pattern of cellular localization to the centrosome after internalization in PSMA(+) PC3-PIP cells. These characteristics make 5D3 mAb an ideal bioligand to deliver tubulin inhibitors, such as mertansine, to the cell centrosome, leading to mitotic arrest and elimination of dividing PC cells. We have successfully developed a 5D3 mAb- and mertansine (DM1)-based antibody-drug conjugate (ADC) and evaluated it in vitro for binding affinity, internalization, and cytotoxicity. The in vivo therapeutic efficacy of 5D3-DM1 ADC was evaluated in PSMA(+) PC3-PIP and PSMA(-) PC3-Flu mouse models of human PC. This therapeutic study has revealed that this new anti-PSMA ADC can successfully control the growth of PSMA(+) tumors without inducing systemic toxicity.
Keywords: 5D3 antibody; MCC linker; anti-PSMA antibody; antibody-drug conjugates (ADC); drug delivery; mertansine (DM1); prostate cancer; prostate-specific membrane antigen (PSMA); targeted therapy.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

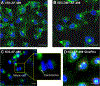

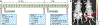


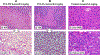

References
-
- Siegel RL; Miller KD; Jemal A Cancer Statistics, 2019. Ca—Cancer J. Clin 2019, 69, 7–34. - PubMed
-
- Siegel RL; Miller KD; Jemal A Cancer Statistics, 2020. Ca—Cancer J. Clin 2020, 70, 7–30. - PubMed
-
- Cho S; Zammarchi F; Williams DG; Havenith CEG; Monks NR; Tyrer P; D’Hooge F; Fleming R; Vashisht K; Dimasi N; et al. Antitumor Activity of Medi3726 (Adct-401), a Pyrrolobenzodiazepine Antibody-Drug Conjugate Targeting Psma, in Preclinical Models of Prostate Cancer. Mol. Cancer Ther 2018, 17, 2176–2186. - PubMed
-
- Phillips RM; Deek MP; Deweese TL; Tran PT Metastasis-Directed Therapy in Prostate Cancer. Why, When, and How? Oncology, 2019, 33, 394–399. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

