Modulating Lipoprotein Transcellular Transport and Atherosclerotic Plaque Formation in ApoE-/- Mice via Nanoformulated Lipid-Methotrexate Conjugates
- PMID: 32805983
- PMCID: PMC7453397
- DOI: 10.1021/acsami.0c12202
Modulating Lipoprotein Transcellular Transport and Atherosclerotic Plaque Formation in ApoE-/- Mice via Nanoformulated Lipid-Methotrexate Conjugates
Abstract
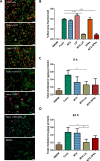
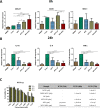
Macrophage inflammation and maturation into foam cells, following the engulfment of oxidized low-density lipoproteins (oxLDL), are major hallmarks in the onset and progression of atherosclerosis. Yet, chronic treatments with anti-inflammatory agents, such as methotrexate (MTX), failed to modulate disease progression, possibly for the limited drug bioavailability and plaque deposition. Here, MTX-lipid conjugates, based on 1,2-distearoyl-sn-glycero-3-phosphoethanolamine (DSPE), were integrated in the structure of spherical polymeric nanoparticles (MTX-SPNs) or intercalated in the lipid bilayer of liposomes (MTX-LIP). Although, both nanoparticles were colloidally stable with an average diameter of ∼200 nm, MTX-LIP exhibited a higher encapsulation efficiency (>70%) and slower release rate (∼50% at 10 h) compared to MTX-SPN. In primary bone marrow derived macrophages (BMDMs), MTX-LIP modulated the transcellular transport of oxLDL more efficiently than free MTX mostly by inducing a 2-fold overexpression of ABCA1 (regulating oxLDL efflux), while the effect on CD36 and SRA-1 (regulating oxLDL influx) was minimal. Furthermore, in BMDMs, MTX-LIP showed a stronger anti-inflammatory activity than free MTX, reducing the expression of IL-1β by 3-fold, IL-6 by 2-fold, and also moderately of TNF-α. In 28 days high-fat-diet-fed apoE-/- mice, MTX-LIP reduced the mean plaque area by 2-fold and the hematic amounts of RANTES by half as compared to free MTX. These results would suggest that the nanoenhanced delivery to vascular plaques of the anti-inflammatory DSPE-MTX conjugate could effectively modulate the disease progression by halting monocytes' maturation and recruitment already at the onset of atherosclerosis.
Keywords: atherosclerosis; foam cells; inflammation; low-density lipoprotein transport; nanomedicine.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



References
-
- Libby P.; Hansson G. K.. Taming Immune and Inflammatory Responses to Treat Atherosclerosis. Journal of the American College of Cardiology 2018. - PubMed
-
- Kim Y.; Lobatto M. E.; Kawahara T.; Lee Chung B.; Mieszawska A. J.; Sanchez-Gaytan B. L.; Fay F.; Senders M. L.; Calcagno C.; Becraft J.; Tun Saung M.; Gordon R. E.; Stroes E. S. G.; Ma M.; Farokhzad O. C.; Fayad Z. A.; Mulder W. J. M.; Langer R. Probing Nanoparticle Translocation across the Permeable Endothelium in Experimental Atherosclerosis. Proc. Natl. Acad. Sci. U. S. A. 2014, 111, 1078–1083. 10.1073/pnas.1322725111. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

