Point-to-Axial Chirality Transmission: A Highly Sensitive Triaryl Chirality Probe for Stereochemical Assignments of Amines
- PMID: 32806087
- PMCID: PMC7458434
- DOI: 10.1021/acs.joc.0c00734
Point-to-Axial Chirality Transmission: A Highly Sensitive Triaryl Chirality Probe for Stereochemical Assignments of Amines
Abstract
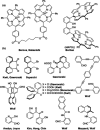
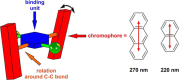
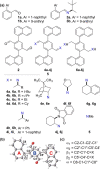
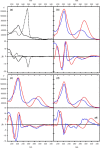
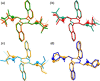
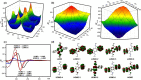
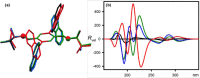
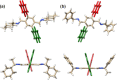
A readily available stereodynamic and the electronic circular dichroism (ECD)-silent 2,5-di(1-naphthyl)-terephthalaldehyde-based probe has been applied for chirality sensing of primary amines. The chiral amine (the inductor) forces a change in the structure of the chromophore system through the point-to-axial chirality transmission mechanism. As a result, efficient induction of optical activity in the chromophoric system is observed. The butterflylike structure of the probe, with the terminal aryl groups acting as changeable "wings", allowed for the generation of exciton Cotton effects in the region of 1Bb electronic transition in the naphthalene chromophores. The sign of the exciton couplets observed for inductor-reporter systems might be correlated with an absolute configuration of the inductor, whereas the linear relationship between amplitudes of the specific Cotton effect and enantiomeric excess of the parent amine gives potentiality for quantitative chirality sensing. Despite the structural simplicity, the probe turned out to be unprecedentedly highly sensitive to even subtle differences in the inductor structure (i.e., O vs CH2).
Conflict of interest statement
The authors declare no competing financial interest.
Figures


Similar articles
-
Optical Activity and Helicity Enhancement of Highly Sensitive Dinaphthylmethane-Based Stereodynamic Probes for Secondary Alcohols.ACS Omega. 2019 Feb 14;4(2):3244-3256. doi: 10.1021/acsomega.8b03337. eCollection 2019 Feb 28. ACS Omega. 2019. PMID: 31459541 Free PMC article.
-
Chirality sensing using stereodynamic probes with distinct electronic circular dichroism output.Chem Soc Rev. 2013 Jun 21;42(12):5408-24. doi: 10.1039/c3cs35498a. Chem Soc Rev. 2013. PMID: 23482984
-
A Helically-Twisted Stereodynamic Probe for Chiroptical Sensing of Chiral Amines through Point-to-Helical Chirality Transmission.Chem Asian J. 2025 Apr 3;20(7):e202401376. doi: 10.1002/asia.202401376. Epub 2025 Jan 15. Chem Asian J. 2025. PMID: 39745148
-
The enduring legacy of Koji Nakanishi's research on natural products and bioorganic chemistry. Part 2. Inception and establishment of the ECD exciton chirality method in 1960s to 1970s: A marvel of Nakanishi's Japanese team.Chirality. 2020 May;32(5):535-546. doi: 10.1002/chir.23193. Epub 2020 Mar 6. Chirality. 2020. PMID: 32142193 Review.
-
ECD exciton chirality method today: a modern tool for determining absolute configurations.Chirality. 2022 Feb;34(2):333-363. doi: 10.1002/chir.23393. Epub 2021 Nov 17. Chirality. 2022. PMID: 34791704 Free PMC article. Review.
References
-
- Berg J. M.; Tymoczko J. L.; Stryer L.. Biochemistry; W. H. Freeman: New York, 2002.
-
- Lough W. J.; Wainer I. W., Eds. Chirality in Natural and Applied Science; CRC Press: Boca Raton, 2002.
-
- Lightner D. A.; Gurst J. E.. Organic Conformational Analysis and Stereochemistry from Circular Dichroism Spectroscopy, Wiley-VCH: New York, 2000.
-
- Eliel E. L.; Wilen S. H.. Stereochemistry of Organic Compounds; John Wiley and Sons Inc.: New York, 1994.
-
- Jacobsen E. N.; Pfaltz A.; Yamamoto H., Eds. Comprehensive Asymmetric Catalysis; Springer: Berlin, Heidelberg, 2004.
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

