When It Comes to an End: Oxidative Stress Crosstalk with Protein Aggregation and Neuroinflammation Induce Neurodegeneration
- PMID: 32806679
- PMCID: PMC7463521
- DOI: 10.3390/antiox9080740
When It Comes to an End: Oxidative Stress Crosstalk with Protein Aggregation and Neuroinflammation Induce Neurodegeneration
Abstract
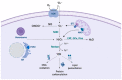
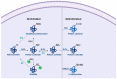
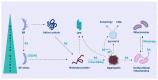
Neurodegenerative diseases are characterized by a progressive loss of neurons in the brain or spinal cord that leads to a loss of function of the affected areas. The lack of effective treatments and the ever-increasing life expectancy is raising the number of individuals affected, having a tremendous social and economic impact. The brain is particularly vulnerable to oxidative damage given the high energy demand, low levels of antioxidant defenses, and high levels of metal ions. Driven by age-related changes, neurodegeneration is characterized by increased oxidative stress leading to irreversible neuronal damage, followed by cell death. Nevertheless, neurodegenerative diseases are known as complex pathologies where several mechanisms drive neuronal death. Herein we discuss the interplay among oxidative stress, proteinopathy, and neuroinflammation at the early stages of neurodegenerative diseases. Finally, we discuss the use of the Nrf2-ARE pathway as a potential therapeutic strategy based on these molecular mechanisms to develop transformative medicines.
Keywords: Nrf2-ARE pathway; neurodegenerative diseases; neuroinflammation; oxidative stress; protein aggregates; reactive species; redox signaling.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

