WNT Signaling in Lung Repair and Regeneration
- PMID: 32807748
- PMCID: PMC7528681
- DOI: 10.14348/molcells.2020.0059
WNT Signaling in Lung Repair and Regeneration
Abstract
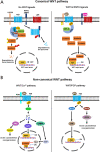
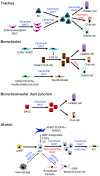
The lung has a vital function in gas exchange between the blood and the external atmosphere. It also has a critical role in the immune defense against external pathogens and environmental factors. While the lung is classified as a relatively quiescent organ with little homeostatic turnover, it shows robust regenerative capacity in response to injury, mediated by the resident stem/progenitor cells. During regeneration, regionally distinct epithelial cell populations with specific functions are generated from several different types of stem/progenitor cells localized within four histologically distinguished regions: trachea, bronchi, bronchioles, and alveoli. WNT signaling is one of the key signaling pathways involved in regulating many types of stem/progenitor cells in various organs. In addition to its developmental role in the embryonic and fetal lung, WNT signaling is critical for lung homeostasis and regeneration. In this minireview, we summarize and discuss recent advances in the understanding of the role of WNT signaling in lung regeneration with an emphasis on stem/progenitor cells.
Keywords: WNT signaling; lung homeostasis; lung regeneration; lung stem/progenitor cells; β-catenin.
Conflict of interest statement
The authors have no potential conflicts of interest to disclose.
Figures


References
-
- Basil M.C., Katzen J., Engler A.E., Guo M., Herriges M.J., Kathiriya J.J., Windmueller R., Ysasi A.B., Zacharias W.J., Chapman H.A., et al. The cellular and physiological basis for lung repair and regeneration: past, present, and future. Cell Stem Cell. 2020;26:482–502. doi: 10.1016/j.stem.2020.03.009. - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

