Multiscale Simulation Reveals Passive Proton Transport Through SERCA on the Microsecond Timescale
- PMID: 32814059
- PMCID: PMC7474205
- DOI: 10.1016/j.bpj.2020.07.027
Multiscale Simulation Reveals Passive Proton Transport Through SERCA on the Microsecond Timescale
Abstract
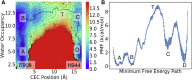
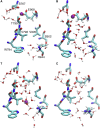
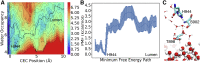
The sarcoplasmic reticulum Ca2+-ATPase (SERCA) transports two Ca2+ ions from the cytoplasm to the reticulum lumen at the expense of ATP hydrolysis. In addition to transporting Ca2+, SERCA facilitates bidirectional proton transport across the sarcoplasmic reticulum to maintain the charge balance of the transport sites and to balance the charge deficit generated by the exchange of Ca2+. Previous studies have shown the existence of a transient water-filled pore in SERCA that connects the Ca2+ binding sites with the lumen, but the capacity of this pathway to sustain passive proton transport has remained unknown. In this study, we used the multiscale reactive molecular dynamics method and free energy sampling to quantify the free energy profile and timescale of the proton transport across this pathway while also explicitly accounting for the dynamically coupled hydration changes of the pore. We find that proton transport from the central binding site to the lumen has a microsecond timescale, revealing a novel passive cytoplasm-to-lumen proton flow beside the well-known inverse proton countertransport occurring in active Ca2+ transport. We propose that this proton transport mechanism is operational and serves as a functional conduit for passive proton transport across the sarcoplasmic reticulum.
Copyright © 2020 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures
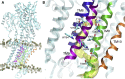
Comment in
-
A Computational Swiss Army Knife Approach to Unraveling the Secrets of Proton Movement through SERCA.Biophys J. 2020 Sep 1;119(5):890-891. doi: 10.1016/j.bpj.2020.07.028. Epub 2020 Aug 6. Biophys J. 2020. PMID: 32795397 Free PMC article. No abstract available.
Similar articles
-
Structural Basis for the Function of the C-Terminal Proton Release Pathway in the Calcium Pump.Int J Mol Sci. 2021 Mar 29;22(7):3507. doi: 10.3390/ijms22073507. Int J Mol Sci. 2021. PMID: 33805255 Free PMC article.
-
The Ca2+-ATPase pump facilitates bidirectional proton transport across the sarco/endoplasmic reticulum.Mol Biosyst. 2017 Mar 28;13(4):633-637. doi: 10.1039/c7mb00065k. Mol Biosyst. 2017. PMID: 28290590 Free PMC article. Review.
-
Microsecond Molecular Simulations Reveal a Transient Proton Pathway in the Calcium Pump.J Am Chem Soc. 2015 Jun 10;137(22):7055-8. doi: 10.1021/jacs.5b03814. Epub 2015 Jun 2. J Am Chem Soc. 2015. PMID: 26028459
-
Proton Countertransport and Coupled Gating in the Sarcoplasmic Reticulum Calcium Pump.J Mol Biol. 2018 Dec 7;430(24):5050-5065. doi: 10.1016/j.jmb.2018.10.014. Epub 2018 Oct 26. J Mol Biol. 2018. PMID: 30539761 Free PMC article.
-
Ion pathways in the sarcoplasmic reticulum Ca2+-ATPase.J Biol Chem. 2013 Apr 12;288(15):10759-65. doi: 10.1074/jbc.R112.436550. Epub 2013 Feb 11. J Biol Chem. 2013. PMID: 23400778 Free PMC article. Review.
Cited by
-
Multiscale Simulation of an Influenza A M2 Channel Mutant Reveals Key Features of Its Markedly Different Proton Transport Behavior.J Am Chem Soc. 2022 Jan 19;144(2):769-776. doi: 10.1021/jacs.1c09281. Epub 2022 Jan 5. J Am Chem Soc. 2022. PMID: 34985907 Free PMC article.
-
Allosteric Modulation of SERCA Pumps in Health and Disease: Structural Dynamics, Posttranslational Modifications, and Therapeutic Potential.J Mol Biol. 2025 May 9:169200. doi: 10.1016/j.jmb.2025.169200. Online ahead of print. J Mol Biol. 2025. PMID: 40349954 Review.
-
A Computational Swiss Army Knife Approach to Unraveling the Secrets of Proton Movement through SERCA.Biophys J. 2020 Sep 1;119(5):890-891. doi: 10.1016/j.bpj.2020.07.028. Epub 2020 Aug 6. Biophys J. 2020. PMID: 32795397 Free PMC article. No abstract available.
-
Structural Basis for the Function of the C-Terminal Proton Release Pathway in the Calcium Pump.Int J Mol Sci. 2021 Mar 29;22(7):3507. doi: 10.3390/ijms22073507. Int J Mol Sci. 2021. PMID: 33805255 Free PMC article.
-
Deactivation blocks proton pathways in the mitochondrial complex I.Proc Natl Acad Sci U S A. 2021 Jul 20;118(29):e2019498118. doi: 10.1073/pnas.2019498118. Proc Natl Acad Sci U S A. 2021. PMID: 34272275 Free PMC article.
References
-
- Levy D., Seigneuret M., Rigaud J.L. Evidence for proton countertransport by the sarcoplasmic reticulum Ca2+-ATPase during calcium transport in reconstituted proteoliposomes with low ionic permeability. J. Biol. Chem. 1990;265:19524–19534. - PubMed
-
- Forge V., Mintz E., Guillain F. Ca2+ binding to sarcoplasmic reticulum ATPase revisited. I. Mechanism of affinity and cooperativity modulation by H+ and Mg2+ J. Biol. Chem. 1993;268:10953–10960. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

