Correlated clusters of closed reaction centers during induction of intact cells of photosynthetic bacteria
- PMID: 32814810
- PMCID: PMC7438532
- DOI: 10.1038/s41598-020-70966-3
Correlated clusters of closed reaction centers during induction of intact cells of photosynthetic bacteria
Abstract
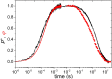
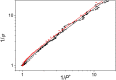
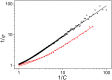
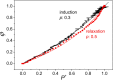
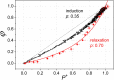
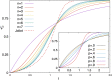
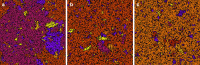
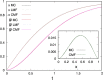
Antenna systems serve to absorb light and to transmit excitation energy to the reaction center (RC) in photosynthetic organisms. As the emitted (bacterio)chlorophyll fluorescence competes with the photochemical utilization of the excitation, the measured fluorescence yield is informed by the migration of the excitation in the antenna. In this work, the fluorescence yield concomitant with the oxidized dimer (P+) of the RC were measured during light excitation (induction) and relaxation (in the dark) for whole cells of photosynthetic bacterium Rhodobacter sphaeroides lacking cytochrome c2 as natural electron donor to P+ (mutant cycA). The relationship between the fluorescence yield and P+ (fraction of closed RC) showed deviations from the standard Joliot-Lavergne-Trissl model: (1) the hyperbola is not symmetric and (2) exhibits hysteresis. These phenomena originate from the difference between the delays of fluorescence relative to P+ kinetics during induction and relaxation, and in structural terms from the non-random distribution of the closed RCs during induction. The experimental findings are supported by Monte Carlo simulations and by results from statistical physics based on random walk approximations of the excitation in the antenna. The applied mathematical treatment demonstrates the generalization of the standard theory and sets the stage for a more adequate description of the long-debated kinetics of fluorescence and of the delicate control and balance between efficient light harvest and photoprotection in photosynthetic organisms.
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Roadmap of electrons from donor side to the reaction center of photosynthetic purple bacteria with mutated cytochromes.Photosynth Res. 2024 Mar;159(2-3):261-272. doi: 10.1007/s11120-023-01059-1. Epub 2023 Nov 30. Photosynth Res. 2024. PMID: 38032488 Free PMC article.
-
Fluorescence relaxation in intact cells of photosynthetic bacteria: donor and acceptor side limitations of reopening of the reaction center.Photosynth Res. 2015 Apr;124(1):31-44. doi: 10.1007/s11120-014-0070-0. Epub 2014 Dec 20. Photosynth Res. 2015. PMID: 25527461
-
The effect of light and temperature on the dynamic state of Rhodobacter sphaeroides reaction centers proteins determined from changes in tryptophan fluorescence lifetime and P+QA- recombination kinetics.J Photochem Photobiol B. 2018 Mar;180:140-148. doi: 10.1016/j.jphotobiol.2018.01.027. Epub 2018 Feb 6. J Photochem Photobiol B. 2018. PMID: 29413697
-
Structural and functional proteomics of intracytoplasmic membrane assembly in Rhodobacter sphaeroides.J Mol Microbiol Biotechnol. 2013;23(1-2):48-62. doi: 10.1159/000346520. Epub 2013 Apr 18. J Mol Microbiol Biotechnol. 2013. PMID: 23615195 Review.
-
The photosynthetic apparatus of Rhodobacter sphaeroides.Trends Microbiol. 1999 Nov;7(11):435-40. doi: 10.1016/s0966-842x(99)01625-x. Trends Microbiol. 1999. PMID: 10542422 Review.
Cited by
-
Roadmap of electrons from donor side to the reaction center of photosynthetic purple bacteria with mutated cytochromes.Photosynth Res. 2024 Mar;159(2-3):261-272. doi: 10.1007/s11120-023-01059-1. Epub 2023 Nov 30. Photosynth Res. 2024. PMID: 38032488 Free PMC article.
-
Capacity and kinetics of light-induced cytochrome oxidation in intact cells of photosynthetic bacteria.Sci Rep. 2022 Aug 22;12(1):14298. doi: 10.1038/s41598-022-18399-y. Sci Rep. 2022. PMID: 35995915 Free PMC article.
-
Probing the design principles of photosynthetic systems through fluorescence noise measurement.Sci Rep. 2024 Jun 16;14(1):13877. doi: 10.1038/s41598-024-64068-7. Sci Rep. 2024. PMID: 38880795 Free PMC article.
-
Radiation-resistant bacteria in desiccated soil and their potentiality in applied sciences.Front Microbiol. 2024 Jun 4;15:1348758. doi: 10.3389/fmicb.2024.1348758. eCollection 2024. Front Microbiol. 2024. PMID: 38894973 Free PMC article. Review.
-
Light-induced reversible reorganizations in closed Type II reaction centre complexes: physiological roles and physical mechanisms.Open Biol. 2022 Dec;12(12):220297. doi: 10.1098/rsob.220297. Epub 2022 Dec 14. Open Biol. 2022. PMID: 36514981 Free PMC article. Review.
References
-
- Mirkovic, T., Ostroumov, E. E., Anna, J. M., van Grondelle, R., Govindjee & Scholes, G. D. Light absorption and energy transfer in the antenna complexes of photosynthetic organisms. Chem. Rev.117(2), 249-293 (2016). - PubMed
-
- Maróti, P. & Govindjee. Energy conversion in photosynthetic bacteria. Photosynth. Res.127(2), 257–271 (2016). - PubMed
-
- Maróti P. Chemical rescue of delivery in proton transfer mutants of reaction center of photosynthetic bacteria. Biochim. Biophys. Acta Bioenerg. 2019;1860:317–324. - PubMed
-
- Maróti P. Thermodynamic view of proton activated electron transfer in the reaction center of photosynthetic bacteria. J. Phys. Chem. B. 2019;123:5463–5473. - PubMed
-
- Franck J, Teller E. Migration and photochemical action of excitation energy in crystals. J. Chem. Phys. 1938;6:861–872.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

