Laminar Subnetworks of Response Suppression in Macaque Primary Visual Cortex
- PMID: 32817246
- PMCID: PMC7511183
- DOI: 10.1523/JNEUROSCI.1129-20.2020
Laminar Subnetworks of Response Suppression in Macaque Primary Visual Cortex
Abstract
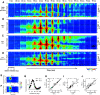
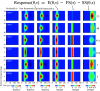
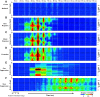
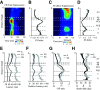
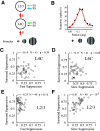
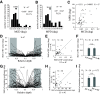
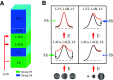
Cortical inhibition plays an important role in information processing in the brain. However, the mechanisms by which inhibition and excitation are coordinated to generate functions in the six layers of the cortex remain unclear. Here, we measured laminar-specific responses to stimulus orientations in primary visual cortex (V1) of awake monkeys (male, Macaca mulatta). We distinguished inhibitory effects (suppression) from excitation, by taking advantage of the separability of excitation and inhibition in the orientation and time domains. We found two distinct types of suppression governing different layers. Fast suppression (FS) was strongest in input layers (4C and 6), and slow suppression (SS) was 3 times stronger in output layers (2/3 and 5). Interestingly, the two types of suppression were correlated with different functional properties measured with drifting gratings. FS was primarily correlated with orientation selectivity in input layers (r = -0.65, p < 10-9), whereas SS was primarily correlated with surround suppression in output layers (r = 0.61, p < 10-4). The earliest SS in layer 1 indicates the origin of cortical feedback for SS, in contrast to the feedforward/recurrent origin of FS. Our results reveal two V1 laminar subnetworks with different response suppression that may provide a general framework for laminar processing in other sensory cortices.SIGNIFICANCE STATEMENT This study sought to understand inhibitory effects (suppression) and their relationships with functional properties in the six different layers of the cortex. We found that the diversity of neural responses across layers in primary visual cortex (V1) could be fully explained by one excitatory and two suppressive components (fast and slow suppression). The distinct laminar distributions, origins, and functional roles of the two types of suppression provided a simplified representation of the differences between two V1 subnetworks (input network and output network). These results not only help to elucidate computational principles in macaque V1, but also provide a framework for general computation of cortical laminae in other sensory cortices.
Keywords: Macaque monkey; cortical layers; neural dynamics; primary visual cortex; suppression.
Copyright © 2020 Wang et al.
Figures





Similar articles
-
Dynamic Recruitment of the Feedforward and Recurrent Mechanism for Black-White Asymmetry in the Primary Visual Cortex.J Neurosci. 2023 Aug 2;43(31):5668-5684. doi: 10.1523/JNEUROSCI.0168-23.2023. Epub 2023 Jul 24. J Neurosci. 2023. PMID: 37487737 Free PMC article.
-
Layer 3 Dynamically Coordinates Columnar Activity According to Spatial Context.J Neurosci. 2019 Jan 9;39(2):281-294. doi: 10.1523/JNEUROSCI.1568-18.2018. Epub 2018 Nov 20. J Neurosci. 2019. PMID: 30459226 Free PMC article.
-
Laminar Differences in Responses to Naturalistic Texture in Macaque V1 and V2.J Neurosci. 2019 Dec 4;39(49):9748-9756. doi: 10.1523/JNEUROSCI.1743-19.2019. Epub 2019 Oct 30. J Neurosci. 2019. PMID: 31666355 Free PMC article.
-
Dynamics of orientation selectivity in the primary visual cortex and the importance of cortical inhibition.Neuron. 2003 Jun 5;38(5):689-99. doi: 10.1016/s0896-6273(03)00332-5. Neuron. 2003. PMID: 12797955 Review.
-
Local circuits in primary visual cortex of the macaque monkey.Annu Rev Neurosci. 1998;21:47-74. doi: 10.1146/annurev.neuro.21.1.47. Annu Rev Neurosci. 1998. PMID: 9530491 Review.
Cited by
-
Coding strategy for surface luminance switches in the primary visual cortex of the awake monkey.Nat Commun. 2022 Jan 12;13(1):286. doi: 10.1038/s41467-021-27892-3. Nat Commun. 2022. PMID: 35022404 Free PMC article.
-
V1-bypassing suppression leads to direction-specific microsaccade modulation in visual coding and perception.Nat Commun. 2022 Oct 26;13(1):6366. doi: 10.1038/s41467-022-34057-3. Nat Commun. 2022. PMID: 36289224 Free PMC article.
-
Temporal characteristics of gamma rhythm constrain properties of noise in an inhibition-stabilized network model.Cereb Cortex. 2023 Sep 9;33(18):10108-10121. doi: 10.1093/cercor/bhad270. Cereb Cortex. 2023. PMID: 37492002 Free PMC article.
-
Dynamic Recruitment of the Feedforward and Recurrent Mechanism for Black-White Asymmetry in the Primary Visual Cortex.J Neurosci. 2023 Aug 2;43(31):5668-5684. doi: 10.1523/JNEUROSCI.0168-23.2023. Epub 2023 Jul 24. J Neurosci. 2023. PMID: 37487737 Free PMC article.
-
Nonuniform and pathway-specific laminar processing of spatial frequencies in the primary visual cortex of primates.Nat Commun. 2024 May 13;15(1):4005. doi: 10.1038/s41467-024-48379-x. Nat Commun. 2024. PMID: 38740786 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
