Rapid and quantitative antimalarial drug efficacy testing via the magneto-optical detection of hemozoin
- PMID: 32820190
- PMCID: PMC7441145
- DOI: 10.1038/s41598-020-70860-y
Rapid and quantitative antimalarial drug efficacy testing via the magneto-optical detection of hemozoin
Abstract
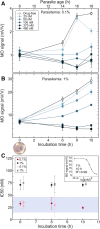
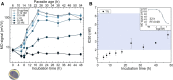
Emergence of resistant Plasmodium species makes drug efficacy testing a crucial part of malaria control. Here we describe a novel assay for sensitive, fast and simple drug screening via the magneto-optical detection of hemozoin, a natural biomarker formed during the hemoglobin metabolism of Plasmodium species. By quantifying hemozoin production over the intraerythrocytic cycle, we reveal that hemozoin formation is already initiated by ~ 6-12 h old ring-stage parasites. We demonstrate that the new assay is capable of drug efficacy testing with incubation times as short as 6-10 h, using synchronized P. falciparum 3D7 cultures incubated with chloroquine, piperaquine and dihydroartemisinin. The determined 50% inhibitory concentrations agree well with values established by standard assays requiring significantly longer testing time. Accordingly, we conclude that magneto-optical hemozoin detection provides a practical approach for the quick assessment of drug effect with short incubation times, which may also facilitate stage-specific assessment of drug inhibitory effects.
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
Magneto-optical assessment of Plasmodium parasite growth via hemozoin crystal size.Sci Rep. 2024 Jun 21;14(1):14318. doi: 10.1038/s41598-024-60988-6. Sci Rep. 2024. PMID: 38906910 Free PMC article.
-
A novel flow cytometric hemozoin detection assay for real-time sensitivity testing of Plasmodium falciparum.PLoS One. 2013 Apr 24;8(4):e61606. doi: 10.1371/journal.pone.0061606. Print 2013. PLoS One. 2013. PMID: 23637865 Free PMC article.
-
Bazedoxifene, a Postmenopausal Drug, Acts as an Antimalarial and Inhibits Hemozoin Formation.Microbiol Spectr. 2022 Jun 29;10(3):e0278121. doi: 10.1128/spectrum.02781-21. Epub 2022 May 26. Microbiol Spectr. 2022. PMID: 35616371 Free PMC article.
-
Antimalarial drugs inhibiting hemozoin (beta-hematin) formation: a mechanistic update.Life Sci. 2007 Feb 6;80(9):813-28. doi: 10.1016/j.lfs.2006.11.008. Epub 2006 Nov 10. Life Sci. 2007. PMID: 17157328 Review.
-
Involvement of heme in the antimalarial action of chloroquine.Trans Am Clin Climatol Assoc. 1998;109:97-105; discussion 105-6. Trans Am Clin Climatol Assoc. 1998. PMID: 9601131 Free PMC article. Review.
Cited by
-
In-silico analysis of potent Mosquirix vaccine adjuvant leads.J Genet Eng Biotechnol. 2023 Nov 30;21(1):155. doi: 10.1186/s43141-023-00590-x. J Genet Eng Biotechnol. 2023. PMID: 38032502 Free PMC article.
-
Sensitive detection of Plasmodium vivax malaria by the rotating-crystal magneto-optical method in Thailand.Sci Rep. 2021 Sep 17;11(1):18547. doi: 10.1038/s41598-021-97532-9. Sci Rep. 2021. PMID: 34535695 Free PMC article.
-
Towards rainbow portable Cytophone with laser diodes for global disease diagnostics.Sci Rep. 2022 May 23;12(1):8671. doi: 10.1038/s41598-022-11452-w. Sci Rep. 2022. PMID: 35606373 Free PMC article.
-
Magneto-optical diagnosis of symptomatic malaria in Papua New Guinea.Nat Commun. 2021 Feb 12;12(1):969. doi: 10.1038/s41467-021-21110-w. Nat Commun. 2021. PMID: 33579923 Free PMC article.
-
Next Generation Chemiluminescent Probes for Antimalarial Drug Discovery.ACS Infect Dis. 2024 Apr 12;10(4):1286-1297. doi: 10.1021/acsinfecdis.3c00707. Epub 2024 Apr 1. ACS Infect Dis. 2024. PMID: 38556981 Free PMC article.
References
-
- Health Organization, W. Artemisinin resistance and artemisinin-based combination therapy efficacy (Status report—August 2018).
-
- Eyasu M. Antimalarial drug resistance: in the past, current status and future perspectives. Br. J. Pharmacol. Toxicol. 2015;6:1–15.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

