Large Scale, Multicenter, Prospective Study of Apatinib in Advanced Gastric Cancer: A Real-World Study from China
- PMID: 32821164
- PMCID: PMC7418160
- DOI: 10.2147/CMAR.S249153
Large Scale, Multicenter, Prospective Study of Apatinib in Advanced Gastric Cancer: A Real-World Study from China
Erratum in
-
Erratum: Large Scale, Multicenter, Prospective Study of Apatinib in Advanced Gastric Cancer: A Real-World Study from China [Corrigendum].Cancer Manag Res. 2020 Sep 11;12:8409. doi: 10.2147/CMAR.S279084. eCollection 2020. Cancer Manag Res. 2020. PMID: 32982434 Free PMC article.
Abstract
Background: In China, gastric cancer (GC) ranks second in incidence and mortality. Over 80% of patients with GC were diagnosed at an advanced stage with poor clinical outcome. Chemotherapy was the mainstream treatment with limited benefit. Apatinib, an inhibitor of targeting vascular endothelial growth factor receptor 2 (VEGFR2), has been approved for third-line treatment of advanced gastric cancer. However, the data of apatinib treatment in the real-world setting are limited. In this real-world study, we aimed to understand the current treatment pattern of apatinib, investigate the effectiveness and safety of apatinib in real-world settings, and explore the potential factors associated with the clinical outcomes.
Methods: This was a prospective, multicenter observational study in a real-world setting. Patients aged ≥18 years with histologic diagnosis of advanced GC were eligible for enrollment. The eligible patients received either apatinib monotherapy or apatinib plus chemotherapy by physician's discretion. Apatinib treatment could be used as first-line, second-line, or third-line and above therapy. The primary endpoint was progression-free survival (PFS). The secondary endpoints were overall survival (OS), ORR, DCR, and safety profile.
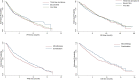
Results: A total of 737 patients with advanced gastric cancer treated with apatinib were included in the FAS population. A total of 54.9% patients used apatinib monotherapy and 45.1% patients used apatinib combination therapy. A total of 44.1% patients received apatinib in first-line treatment, 28.2% in second-line, and 27.7% in third-line and above. In first-line treatment, the objective response rate (ORR) was 9.09% and 16.42% in apatinib monotherapy and combination therapy groups, and disease control rate (DCR) was 78.41% and 89.29%, respectively. Patients who received combination therapy achieved significantly longer median progression-free survival (mPFS; 6.18 vs 3.52 months, p<0.01) and median overall survival (mOS; 8.72 vs 5.92 months, p<0.01) compared with monotherapy. In second-line and third-line therapy, combination therapy showed a better trend in tumor response and survival outcomes compared with monotherapy. For all patients, apatinib combined with paclitaxel were associated with longer mPFS compared with other combinations (8.88 vs 6.62 months). Multivariate analysis showed that combination with paclitaxel (p=0.02) and experience of apatinib-related specific AEs (p<0.01) were independent predictors for PFS and OS. The safety profile was tolerable and no unexpected adverse events were reported.
Conclusion: In a real-world setting, apatinib showed a favorable effectiveness and safety profile in patients with advanced gastric cancer. Apatinib combination therapy, especially combined with paclitaxel, might lead to better survival benefit in first-line treatment. Combination with paclitaxel and the occurrence of apatinib-specific AEs were independent factors associated with better survival outcomes.
Trial registration: NCT03333967.
Keywords: advanced gastric cancer; apatinib; combination therapy; real-world.
© 2020 Peng et al.
Conflict of interest statement
The authors report no conflicts of interest in this work.
Figures
Similar articles
-
Efficacy and safety of second-line therapy with apatinib combined with chemotherapy as second-line therapy in advanced gastric cancer: a single-arm, open-label, prospective, multicenter study.Ann Transl Med. 2022 Jun;10(11):641. doi: 10.21037/atm-22-2752. Ann Transl Med. 2022. PMID: 35813347 Free PMC article.
-
Effectiveness and safety of low-dose apatinib in advanced gastric cancer: A real-world study.Cancer Med. 2020 Jul;9(14):5008-5014. doi: 10.1002/cam4.3105. Epub 2020 May 22. Cancer Med. 2020. PMID: 32441892 Free PMC article.
-
High DCR and Better Survival in Patients with Advanced or Metastatic Gastric Cancer Receiving Anti-Angiogenic TKI plus Chemotherapy: A Real-World Study.Technol Cancer Res Treat. 2023 Jan-Dec;22:15330338221150561. doi: 10.1177/15330338221150561. Technol Cancer Res Treat. 2023. PMID: 36632666 Free PMC article.
-
Comparison of the efficacy and safety of third-line treatments for advanced gastric cancer: A systematic review and network meta-analysis.Front Oncol. 2023 Mar 1;13:1118820. doi: 10.3389/fonc.2023.1118820. eCollection 2023. Front Oncol. 2023. PMID: 36937403 Free PMC article.
-
The safety of apatinib for the treatment of gastric cancer.Expert Opin Drug Saf. 2018 Nov;17(11):1145-1150. doi: 10.1080/14740338.2018.1535592. Epub 2018 Oct 24. Expert Opin Drug Saf. 2018. PMID: 30324820 Review.
Cited by
-
Regulation of regulatory T cells and tumor-associated macrophages in gastric cancer tumor microenvironment.Cancer Med. 2024 Jan;13(2):e6959. doi: 10.1002/cam4.6959. Cancer Med. 2024. PMID: 38349050 Free PMC article. Review.
-
TWIST1-EP300 Expedites Gastric Cancer Cell Resistance to Apatinib by Activating the Expression of COL1A2.Anal Cell Pathol (Amst). 2022 Feb 22;2022:5374262. doi: 10.1155/2022/5374262. eCollection 2022. Anal Cell Pathol (Amst). 2022. PMID: 35242497 Free PMC article.
-
Abnormal phenotype of Nrf2 is associated with poor prognosis through hypoxic/VEGF-A-Rap1b/VEGFR2 pathway in gastric cancer.Aging (Albany NY). 2022 Apr 13;14(7):3293-3312. doi: 10.18632/aging.204013. Epub 2022 Apr 13. Aging (Albany NY). 2022. PMID: 35417854 Free PMC article.
-
Efficacy and Response Biomarkers of Apatinib in the Treatment of Malignancies in China: A Review.Front Oncol. 2021 Oct 5;11:749083. doi: 10.3389/fonc.2021.749083. eCollection 2021. Front Oncol. 2021. PMID: 34676173 Free PMC article. Review.
-
Clinical progress of anti-angiogenic targeted therapy and combination therapy for gastric cancer.Front Oncol. 2023 Jun 12;13:1148131. doi: 10.3389/fonc.2023.1148131. eCollection 2023. Front Oncol. 2023. PMID: 37384288 Free PMC article. Review.
References
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous