Evaluation of seasonal malaria chemoprevention in two areas of intense seasonal malaria transmission: Secondary analysis of a household-randomised, placebo-controlled trial in Houndé District, Burkina Faso and Bougouni District, Mali
- PMID: 32822362
- PMCID: PMC7442230
- DOI: 10.1371/journal.pmed.1003214
Evaluation of seasonal malaria chemoprevention in two areas of intense seasonal malaria transmission: Secondary analysis of a household-randomised, placebo-controlled trial in Houndé District, Burkina Faso and Bougouni District, Mali
Abstract
Background: Seasonal malaria chemoprevention (SMC) is now widely deployed in the Sahel, including several countries that are major contributors to the global burden of malaria. Consequently, it is important to understand whether SMC continues to provide a high level of protection and how SMC might be improved. SMC was evaluated using data from a large, household-randomised trial in Houndé, Burkina Faso and Bougouni, Mali.
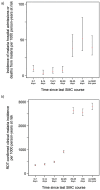
Methods and findings: The parent trial evaluated monthly SMC plus either azithromycin (AZ) or placebo, administered as directly observed therapy 4 times per year between August and November (2014-2016). In July 2014, 19,578 children aged 3-59 months were randomised by household to study group. Children who remained within the age range 3-59 months in August each year, plus children born into study households or who moved into the study area, received study drugs in 2015 and 2016. These analyses focus on the approximately 10,000 children (5,000 per country) under observation each year in the SMC plus placebo group. Despite high coverage and high adherence to SMC, the incidence of hospitalisations or deaths due to malaria and uncomplicated clinical malaria remained high in the study areas (overall incidence rates 12.5 [95% confidence interval (CI): 11.2, 14.1] and 871.1 [95% CI: 852.3, 890.6] cases per 1,000 person-years, respectively) and peaked in July each year, before SMC delivery began in August. The incidence rate ratio comparing SMC within the past 28 days with SMC more than 35 days ago-adjusted for age, country, and household clustering-was 0.13 (95% CI: 0.08, 0.20), P < 0.001 for malaria hospitalisations and deaths from malaria and 0.21 (95% CI 0.20, 0.23), P < 0.001 for uncomplicated malaria, indicating protective efficacy of 87.4% (95% CI: 79.6%, 92.2%) and 78.3% (95% CI: 76.8%, 79.6%), respectively. The prevalence of malaria parasitaemia at weekly surveys during the rainy season and at the end of the transmission season was several times higher in children who missed the SMC course preceding the survey contact, and the smallest prevalence ratio observed was 2.98 (95% CI: 1.95, 4.54), P < 0.001. The frequency of molecular markers of sulfadoxine-pyrimethamine (SP) and amodiaquine (AQ) resistance did not increase markedly over the study period either amongst study children or amongst school-age children resident in the study areas. After 3 years of SMC deployment, the day 28 PCR-unadjusted adequate clinical and parasitological response rate of the SP + AQ regimen in children with asymptomatic malaria was 98.3% (95% CI: 88.6%, 99.8%) in Burkina Faso and 96.1% (95% CI: 91.5%, 98.2%) in Mali. Key limitations of this study are the potential overdiagnosis of uncomplicated malaria by rapid diagnostic tests and the potential for residual confounding from factors related to adherence to the monthly SMC schedule.
Conclusion: Despite strong evidence that SMC is providing a high level of protection, the burden of malaria remains substantial in the 2 study areas. These results emphasise the need for continuing support of SMC programmes. A fifth monthly SMC course is needed to adequately cover the whole transmission season in the study areas and in settings with similar epidemiology.
Trial registration: The AZ-SMC trial in which these data were collected was registered at clinicaltrials.gov: NCT02211729.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


References
-
- Dicko A, Diallo AI, Tembine I, Dicko Y, Dara N, Sidibe Y, et al. Intermittent Preventive Treatment of Malaria Provides Substantial Protection against Malaria in Children Already Protected by an Insecticide-Treated Bednet in Mali: A Randomised, Double-Blind, Placebo-Controlled Trial. PLoS Med. 2011;8(2):e1000407 10.1371/journal.pmed.1000407 - DOI - PMC - PubMed
-
- Konate AT, Yaro JB, Ouedraogo AZ, Diarra A, Gansane A, Soulama I, et al. Intermittent Preventive Treatment of Malaria Provides Substantial Protection against Malaria in Children Already Protected by an Insecticide-Treated Bednet in Burkina Faso: A Randomised, Double-Blind, Placebo-Controlled Trial. PLoS Med. 2011;8(2):e1000408 10.1371/journal.pmed.1000408 - DOI - PMC - PubMed
-
- World Health Organisation. WHO policy recommendation: Seasonal malaria chemoprevention (SMC) for Plasmodium falciparum malaria control in highly seasonal transmission areas of the Sahel sub-region in Africa [Internet]. 2012 [cited 2014 28th May]. http://www.who.int/malaria/publications/atoz/who_smc_policy_recommendati...
-
- World Health Organisation. World Malaria Report 2018. Geneva: World Health Organisation; 2018.
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

