Ethylene Differentially Modulates Hypoxia Responses and Tolerance across Solanum Species
- PMID: 32823611
- PMCID: PMC7465973
- DOI: 10.3390/plants9081022
Ethylene Differentially Modulates Hypoxia Responses and Tolerance across Solanum Species
Abstract
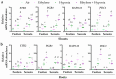
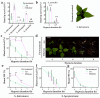
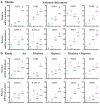
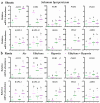
The increasing occurrence of floods hinders agricultural crop production and threatens global food security. The majority of vegetable crops are highly sensitive to flooding and it is unclear how these plants use flooding signals to acclimate to impending oxygen deprivation (hypoxia). Previous research has shown that the early flooding signal ethylene augments hypoxia responses and improves survival in Arabidopsis. To unravel how cultivated and wild Solanum species integrate ethylene signaling to control subsequent hypoxia acclimation, we studied the transcript levels of a selection of marker genes, whose upregulation is indicative of ethylene-mediated hypoxia acclimation in Arabidopsis. Our results suggest that ethylene-mediated hypoxia acclimation is conserved in both shoots and roots of the wild Solanum species bittersweet (Solanum dulcamara) and a waterlogging-tolerant potato (Solanum tuberosum) cultivar. However, ethylene did not enhance the transcriptional hypoxia response in roots of a waterlogging-sensitive potato cultivar, suggesting that waterlogging tolerance in potato could depend on ethylene-controlled hypoxia responses in the roots. Finally, we show that ethylene rarely enhances hypoxia-adaptive genes and does not improve hypoxia survival in tomato (Solanum lycopersicum). We conclude that analyzing genes indicative of ethylene-mediated hypoxia acclimation is a promising approach to identifying key signaling cascades that confer flooding tolerance in crops.
Keywords: PRT6 N-degron pathway of proteolysis; Solanum dulcamara; Solanum lycopersicum; Solanum tuberosum; VII Ethylene Response Factor; ethylene; flooding; hypoxia; phytoglobin.
Conflict of interest statement
The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures


Similar articles
-
Rapid flooding-induced adventitious root development from preformed primordia in Solanum dulcamara.AoB Plants. 2014 Feb 9;6(0):plt058. doi: 10.1093/aobpla/plt058. Print 2014. AoB Plants. 2014. PMID: 24790121 Free PMC article.
-
An overview of low-oxygen sensing and flooding responses of tomato.J Exp Bot. 2025 May 15:eraf203. doi: 10.1093/jxb/eraf203. Online ahead of print. J Exp Bot. 2025. PMID: 40373065
-
Looking for Resistance to Soft Rot Disease of Potatoes Facing Environmental Hypoxia.Int J Mol Sci. 2024 Mar 28;25(7):3757. doi: 10.3390/ijms25073757. Int J Mol Sci. 2024. PMID: 38612570 Free PMC article. Review.
-
The Central Negative Regulator of Flooding Tolerance, the PROTEOLYSIS 6 Branch of the N-degron Pathway, Adversely Modulates Salinity Tolerance in Arabidopsis.Plants (Basel). 2020 Oct 23;9(11):1415. doi: 10.3390/plants9111415. Plants (Basel). 2020. PMID: 33113884 Free PMC article.
-
Flooding tolerance: O2 sensing and survival strategies.Curr Opin Plant Biol. 2013 Oct;16(5):647-53. doi: 10.1016/j.pbi.2013.06.008. Epub 2013 Jul 5. Curr Opin Plant Biol. 2013. PMID: 23830867 Review.
Cited by
-
Low-oxygen-induced root bending is altered by phytoglobin1 through mediation of ethylene response factors (ERFs) and auxin signaling.Planta. 2024 Jul 16;260(2):54. doi: 10.1007/s00425-024-04482-3. Planta. 2024. PMID: 39012577
-
Waterlogging Stress Induces Antioxidant Defense Responses, Aerenchyma Formation and Alters Metabolisms of Banana Plants.Plants (Basel). 2022 Aug 5;11(15):2052. doi: 10.3390/plants11152052. Plants (Basel). 2022. PMID: 35956531 Free PMC article.
-
Hydrogen Sulfide Enhances Plant Tolerance to Waterlogging Stress.Plants (Basel). 2021 Sep 16;10(9):1928. doi: 10.3390/plants10091928. Plants (Basel). 2021. PMID: 34579462 Free PMC article. Review.
-
Identification of ABA-Mediated Genetic and Metabolic Responses to Soil Flooding in Tomato (Solanum lycopersicum L. Mill).Front Plant Sci. 2021 Mar 5;12:613059. doi: 10.3389/fpls.2021.613059. eCollection 2021. Front Plant Sci. 2021. PMID: 33746996 Free PMC article.
-
Reciprocal modulation of responses to nitrate starvation and hypoxia in roots and leaves of Arabidopsis thaliana.Plant Signal Behav. 2024 Dec 31;19(1):2300228. doi: 10.1080/15592324.2023.2300228. Epub 2024 Jan 2. Plant Signal Behav. 2024. PMID: 38165809 Free PMC article.
References
-
- Hirabayashi Y., Mahendran R., Koirala S., Konoshima L., Yamazaki D., Watanabe S., Kim H., Kanae S. Global flood risk under climate change. Nat. Clim. Chang. 2013;3:816–821. doi: 10.1038/nclimate1911. - DOI
-
- Mustroph A. Improving Flooding Tolerance of Crop Plants. Agronomy. 2018;8:160. doi: 10.3390/agronomy8090160. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

