C. elegans Models to Study the Propagation of Prions and Prion-Like Proteins
- PMID: 32824215
- PMCID: PMC7464663
- DOI: 10.3390/biom10081188
C. elegans Models to Study the Propagation of Prions and Prion-Like Proteins
Abstract
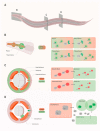
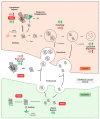
A hallmark common to many age-related neurodegenerative diseases, such as Alzheimer's disease (AD), Parkinson's disease (PD), and amyotrophic lateral sclerosis (ALS), is that patients develop proteinaceous deposits in their central nervous system (CNS). The progressive spreading of these inclusions from initially affected sites to interconnected brain areas is reminiscent of the behavior of bona fide prions in transmissible spongiform encephalopathies (TSEs), hence the term prion-like proteins has been coined. Despite intensive research, the exact mechanisms that facilitate the spreading of protein aggregation between cells, and the associated loss of neurons, remain poorly understood. As population demographics in many countries continue to shift to higher life expectancy, the incidence of neurodegenerative diseases is also rising. This represents a major challenge for healthcare systems and patients' families, since patients require extensive support over several years and there is still no therapy to cure or stop these diseases. The model organism Caenorhabditis elegans offers unique opportunities to accelerate research and drug development due to its genetic amenability, its transparency, and the high degree of conservation of molecular pathways. Here, we will review how recent studies that utilize this soil dwelling nematode have proceeded to investigate the propagation and intercellular transmission of prions and prion-like proteins and discuss their relevance by comparing their findings to observations in other model systems and patients.
Keywords: C. elegans; intercellular spreading; neurodegenerative diseases; prion-like propagation; prions.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Investigating the spreading and toxicity of prion-like proteins using the metazoan model organism C. elegans.J Vis Exp. 2015 Jan 8;(95):52321. doi: 10.3791/52321. J Vis Exp. 2015. PMID: 25591151 Free PMC article.
-
Spreading of a prion domain from cell-to-cell by vesicular transport in Caenorhabditis elegans.PLoS Genet. 2013 Mar;9(3):e1003351. doi: 10.1371/journal.pgen.1003351. Epub 2013 Mar 28. PLoS Genet. 2013. PMID: 23555277 Free PMC article.
-
Synthetic prions and other human neurodegenerative proteinopathies.Virus Res. 2015 Sep 2;207:25-37. doi: 10.1016/j.virusres.2014.10.020. Epub 2014 Oct 31. Virus Res. 2015. PMID: 25449570 Review.
-
[Can prion-like propagation occur in neurodegenerative diseases?: in view of transmissible systemic amyloidosis].Brain Nerve. 2012 Jun;64(6):665-74. Brain Nerve. 2012. PMID: 22647474 Review. Japanese.
-
Insights into Mechanisms of Transmission and Pathogenesis from Transgenic Mouse Models of Prion Diseases.Methods Mol Biol. 2017;1658:219-252. doi: 10.1007/978-1-4939-7244-9_16. Methods Mol Biol. 2017. PMID: 28861793 Free PMC article. Review.
Cited by
-
The role of wild-type tau in Alzheimer's disease and related tauopathies.J Life Sci (Westlake Village). 2020 Dec;2(4):1-17. doi: 10.36069/jols/20201201. J Life Sci (Westlake Village). 2020. PMID: 33665646 Free PMC article.
-
Noncanonical inheritance of phenotypic information by protein amyloids.Nat Cell Biol. 2024 Oct;26(10):1712-1724. doi: 10.1038/s41556-024-01494-9. Epub 2024 Sep 2. Nat Cell Biol. 2024. PMID: 39223373
-
Caenorhabditis elegans Models to Investigate the Mechanisms Underlying Tau Toxicity in Tauopathies.Brain Sci. 2020 Nov 11;10(11):838. doi: 10.3390/brainsci10110838. Brain Sci. 2020. PMID: 33187241 Free PMC article. Review.
-
Modelling the functional genomics of Parkinson's disease in Caenorhabditis elegans: LRRK2 and beyond.Biosci Rep. 2021 Sep 30;41(9):BSR20203672. doi: 10.1042/BSR20203672. Biosci Rep. 2021. PMID: 34397087 Free PMC article. Review.
-
Unraveling Molecular Targets for Neurodegenerative Diseases Through Caenorhabditis elegans Models.Int J Mol Sci. 2025 Mar 26;26(7):3030. doi: 10.3390/ijms26073030. Int J Mol Sci. 2025. PMID: 40243699 Free PMC article. Review.
References
-
- Pan K.M., Baldwin M., Nguyen J., Gasset M., Serban A., Groth D., Mehlhorn I., Huang Z., Fletterick R.J., Cohen F.E., et al. Conversion of alpha-helices into beta-sheets features in the formation of the scrapie prion proteins. Proc. Natl. Acad. Sci. USA. 1993;90:10962–10966. doi: 10.1073/pnas.90.23.10962. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

