Finding specificity in structural brain alterations through Bayesian reverse inference
- PMID: 32829507
- PMCID: PMC7502845
- DOI: 10.1002/hbm.25105
Finding specificity in structural brain alterations through Bayesian reverse inference
Abstract
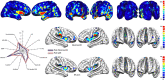
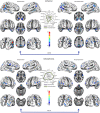
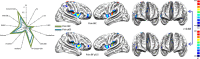
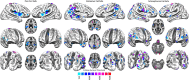
In the field of neuroimaging reverse inferences can lead us to suppose the involvement of cognitive processes from certain patterns of brain activity. However, the same reasoning holds if we substitute "brain activity" with "brain alteration" and "cognitive process" with "brain disorder." The fact that different brain disorders exhibit a high degree of overlap in their patterns of structural alterations makes forward inference-based analyses less suitable for identifying brain areas whose alteration is specific to a certain pathology. In the forward inference-based analyses, in fact, it is impossible to distinguish between areas that are altered by the majority of brain disorders and areas that are specifically affected by certain diseases. To address this issue and allow the identification of highly pathology-specific altered areas we used the Bayes' factor technique, which was employed, as a proof of concept, on voxel-based morphometry data of schizophrenia and Alzheimer's disease. This technique allows to calculate the ratio between the likelihoods of two alternative hypotheses (in our case, that the alteration of the voxel is specific for the brain disorder under scrutiny or that the alteration is not specific). We then performed temporal simulations of the alterations' spread associated with different pathologies. The Bayes' factor values calculated on these simulated data were able to reveal that the areas, which are more specific to a certain disease, are also the ones to be early altered. This study puts forward a new analytical instrument capable of innovating the methodological approach to the investigation of brain pathology.
Keywords: Alzheimer's disease; Bayes' factor; alteration specificity; brain disorders; pain; reverse probability; schizophrenia; voxel-based morphometry.
© 2020 The Authors. Human Brain Mapping published by Wiley Periodicals LLC.
Conflict of interest statement
The authors report no competing interests.
Figures

References
-
- American Psychiatric Association . (2013). Diagnostic and statistical manual of mental disorders, fifth edition (DSM‐5) (5th, ed.). Arlington, VA: American Psychiatric Publishing.

