SARS-CoV-2-specific T cell responses and correlations with COVID-19 patient predisposition
- PMID: 32833687
- PMCID: PMC7685725
- DOI: 10.1172/JCI140965
SARS-CoV-2-specific T cell responses and correlations with COVID-19 patient predisposition
Abstract
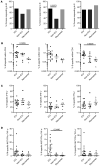
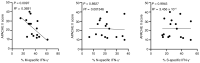
Coronavirus disease 2019 (COVID-19) has emerged as a global pandemic caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). So far, viral targets of cellular immunity and factors determining successful mounting of T cell responses are poorly defined. We therefore analyzed cellular responses to membrane, nucleocapsid, and spike proteins in individuals suffering from moderate or severe infection and in individuals who recovered from mild disease. We demonstrate that the CoV-2-specific CD4+ T helper cell response is directed against all 3 proteins with comparable magnitude, ex vivo proliferation, and portions of responding patients. However, individuals who died were more likely to have not mounted a cellular response to the proteins. Higher patient age and comorbidity index correlated with increased frequencies of CoV-2-specific CD4+ T cells, harboring higher portions of IL-2-secreting, but lower portions of IFN-γ-secreting, cells. Diminished frequencies of membrane protein-reactive IFN-γ+ T cells were particularly associated with higher acute physiology and chronic health evaluation II scores in patients admitted to intensive care. CoV-2-specific T cells exhibited elevated PD-1 expression in patients with active disease as compared with those individuals who recovered from previous mild disease. In summary, our data suggest a link between individual patient predisposition with respect to age and comorbidity and impairment of CoV-2-specific Th1-type cellular immunity, thereby supporting a concept of altered T cell function in at-risk patients.
Keywords: Antigen; COVID-19; T cells.
Conflict of interest statement
Figures





Comment in
-
Are T cells helpful for COVID-19: the relationship between response and risk.J Clin Invest. 2020 Dec 1;130(12):6222-6224. doi: 10.1172/JCI142081. J Clin Invest. 2020. PMID: 32976117 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

