Multimodal brain-age prediction and cardiovascular risk: The Whitehall II MRI sub-study
- PMID: 32835819
- PMCID: PMC8121758
- DOI: 10.1016/j.neuroimage.2020.117292
Multimodal brain-age prediction and cardiovascular risk: The Whitehall II MRI sub-study
Abstract
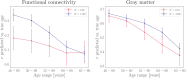
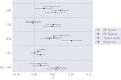
Brain age is becoming a widely applied imaging-based biomarker of neural aging and potential proxy for brain integrity and health. We estimated multimodal and modality-specific brain age in the Whitehall II (WHII) MRI cohort using machine learning and imaging-derived measures of gray matter (GM) morphology, white matter microstructure (WM), and resting state functional connectivity (FC). The results showed that the prediction accuracy improved when multiple imaging modalities were included in the model (R2 = 0.30, 95% CI [0.24, 0.36]). The modality-specific GM and WM models showed similar performance (R2 = 0.22 [0.16, 0.27] and R2 = 0.24 [0.18, 0.30], respectively), while the FC model showed the lowest prediction accuracy (R2 = 0.002 [-0.005, 0.008]), indicating that the FC features were less related to chronological age compared to structural measures. Follow-up analyses showed that FC predictions were similarly low in a matched sub-sample from UK Biobank, and although FC predictions were consistently lower than GM predictions, the accuracy improved with increasing sample size and age range. Cardiovascular risk factors, including high blood pressure, alcohol intake, and stroke risk score, were each associated with brain aging in the WHII cohort. Blood pressure showed a stronger association with white matter compared to gray matter, while no differences in the associations of alcohol intake and stroke risk with these modalities were observed. In conclusion, machine-learning based brain age prediction can reduce the dimensionality of neuroimaging data to provide meaningful biomarkers of individual brain aging. However, model performance depends on study-specific characteristics including sample size and age range, which may cause discrepancies in findings across studies.
Keywords: Brain age prediction; Cardiovascular risk; Machine learning; Multimodal MRI.
Copyright © 2020 The Author(s). Published by Elsevier Inc. All rights reserved.
Figures

References
-
- Beckmann C.F., Mackay C.E., Filippini N., Smith S.M. Group comparison of resting-state FMRI data using multi-subject ICA and dual regression. Neuroimage. 2009;47(Suppl 1):S148.
-
- Beckmann C.F., Smith S.M. Probabilistic independent component analysis for functional magnetic resonance imaging. IEEE Trans. Med. Imaging. 2004;23(2):137–152. - PubMed
-
- Benjamini Y., Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc.: Series B (Methodol.) 1995;57(1):289–300.
Publication types
MeSH terms
Grants and funding
- HQR00984/DH_/Department of Health/United Kingdom
- 203139/Z/16/Z/WT_/Wellcome Trust/United Kingdom
- RF1 AG062553/AG/NIA NIH HHS/United States
- J-0901/PUK_/Parkinson's UK/United Kingdom
- MR/S011676/1/MRC_/Medical Research Council/United Kingdom
- MR/R024790/2/MRC_/Medical Research Council/United Kingdom
- G1001354/MRC_/Medical Research Council/United Kingdom
- MR/R024227/1/MRC_/Medical Research Council/United Kingdom
- S011676/MRC_/Medical Research Council/United Kingdom
- MR/R024790/1/MRC_/Medical Research Council/United Kingdom
- MR/L023784/2/MRC_/Medical Research Council/United Kingdom
- MR/K013351/1/MRC_/Medical Research Council/United Kingdom
- WT_/Wellcome Trust/United Kingdom
- R024227/MRC_/Medical Research Council/United Kingdom
LinkOut - more resources
Full Text Sources
Medical

